

| m(HCl) |
| m(HCl��Һ) |
| m(HCl) |
| 198.25-m(zn) |
| 65 |
| x |
| 2 |
| 0.5g |
| 50.00g-16.25g |
| 50.00g |
| m(HCl) |
| m(HCl��Һ) |
| m(HCl) |
| 198.25-m(zn) |



| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
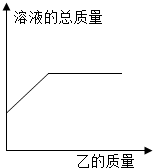 ��2013?�γ�ģ�⣩��һ�������ļ���Һ����������������ʵ���������Һ��������������ҵ�������ϵ��������ͼ���߱�ʾ���� �������� ��2013?�γ�ģ�⣩��һ�������ļ���Һ����������������ʵ���������Һ��������������ҵ�������ϵ��������ͼ���߱�ʾ���� ��������
|
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
 ��2013?�γ�ģ�⣩ʵ���ҳ��ñ��������������Ȼ����Һ��Ӧ��ȡ�����ĵ�������Ӧ�Ļ�ѧ����ʽΪ��NaNO2+NH4Cl=NaCl+N2+2H2O���˷�Ӧ�Ƿ��ȷ�Ӧ����ʵ��װ����ͼ��ʾ���Իش�
��2013?�γ�ģ�⣩ʵ���ҳ��ñ��������������Ȼ����Һ��Ӧ��ȡ�����ĵ�������Ӧ�Ļ�ѧ����ʽΪ��NaNO2+NH4Cl=NaCl+N2+2H2O���˷�Ӧ�Ƿ��ȷ�Ӧ����ʵ��װ����ͼ��ʾ���Իش��鿴�𰸺ͽ���>>
����ѧУ��ѡ - ��ϰ���б� - �����б�
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com