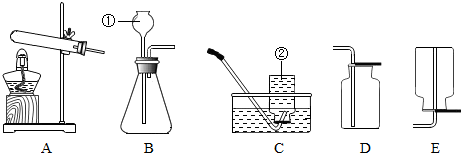
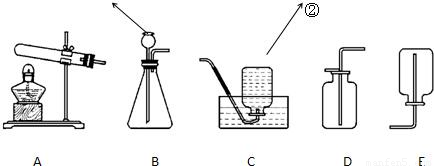
��1��ʵ�����г�������غͶ������̼�����������п����ϡ���ᷴӦ��������ʯ��ʯ��ϡ�����ƶ�����̼���壮
д��������������Ƣ�______ ��______
��������ҩƷѡ���ʵ������巢�����ռ�װ�������±���������ĸ��
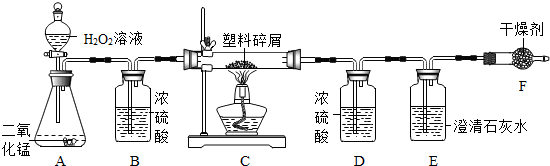
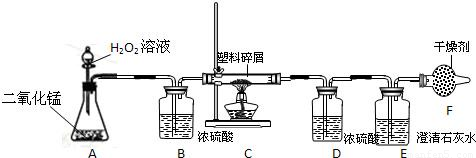
��2�����ڴ���ʹ��һ�������Ϸ������ɵġ���ɫ��Ⱦ�����ѳ�Ϊһ�����ص�������⣮ij��ѧ�о�С���ͬѧ����ij�����ϴ�����ɽ��з���̽����������ʾ������ֻ��C��H����Ԫ�أ��������������ͼ��ʾ��ʵ��װ�ã�ʹ�����������ڴ�����ȼ�գ��۲�ʵ���������й����ݣ�����Ԫ�غ�����
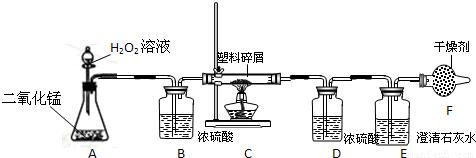
������B��������______��
������E�������______��
��������C�IJ������з����������������ΪWg�������������ȼ�պ�������D����a g����Wg�����������к���Ԫ�ص�����Ϊ______g����������Ϊ������ʽ��
����װ����û����������B����ʹ��������������Ԫ�ص�����������______���ƫС������ƫ������Ӱ�족��֮һ����



 ag��
ag�� a����ƫ��
a����ƫ��

 ����ѧ��ţ��Ӣ��ϵ�д�
����ѧ��ţ��Ӣ��ϵ�д�