
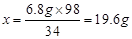 ....��1�֣�
....��1�֣�

 ȫ�ܲ����ĩС״Ԫϵ�д�
ȫ�ܲ����ĩС״Ԫϵ�д�
| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
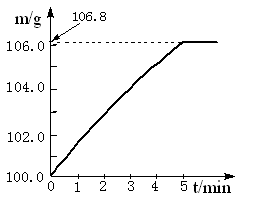
��09���죩ijͬѧΪ�˼�����ﹺ�������炙����Ƿ�ϸ�����ȡ27.5 g������Ʒ������Ũ�ռ���Һ���ȣ������İ�����100.0g���������ա�������հ�������Һ������m�뷴Ӧʱ��t�ı仯������ͼ��ʾ�����漰�ķ�ӦΪ��
(NH4)2SO4+2NaOH��Na2SO4+2H2O+2NH3���� 2NH3+H2SO4 == (NH4)2SO4��
����㣺
��1����ȫ��Ӧ��������� g��
��2���û��ʵĺ�����Ϊ ����ȷ��0.1%����
�����ֻ������� (����ϸ��ϸ�
�ϸ�����狀�����Ϊ20%����)��Ʒ��
��3����������������������������������
���̣���
��4����ʵ������а�������ȫ���գ�����ʵ��������炙��ʵĺ���������ʵ��ֵ����ԭ���� ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2009��ȫ���п���ѧ����������11 ��ѧ����ʽ������ ���ͣ�������
��09���죩ijͬѧΪ�˼�����ﹺ�������炙����Ƿ�ϸ�����ȡ27.5 g������Ʒ������Ũ�ռ���Һ���ȣ������İ�����100.0g���������ա�������հ�������Һ������m�뷴Ӧʱ��t�ı仯������ͼ��ʾ�����漰�ķ�ӦΪ��
(NH4)2SO4+2NaOH��Na2SO4+2H2O+2NH3���� 2NH3+H2SO4 ==(NH4)2SO4��
����㣺
��1����ȫ��Ӧ��������� g��
��2���û��ʵĺ�����Ϊ ����ȷ��0.1%����
�����ֻ������� (����ϸ��ϸ�
�ϸ�����狀�����Ϊ20%����)��Ʒ��
��3����������������������������������
���̣���
��4����ʵ������а�������ȫ���գ�����ʵ��������炙��ʵĺ���������ʵ��ֵ����ԭ���� ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2009��ȫ���п�����ר����ר������ѧ����ʽ���� ���ͣ�������
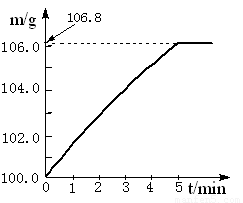
��09���죩ijͬѧΪ�˼�����ﹺ�������炙����Ƿ�ϸ�����ȡ27.5 g������Ʒ������Ũ�ռ���Һ���ȣ������İ�����100.0g���������ա�������հ�������Һ������m�뷴Ӧʱ��t�ı仯������ͼ��ʾ�����漰�ķ�ӦΪ��
(NH4)2SO4+2NaOH��Na2SO4+2H2O+2NH3���� 2NH3+H2SO4 == (NH4)2SO4 ��
����㣺
��1����ȫ��Ӧ��������� g��
��2���û��ʵĺ�����Ϊ ����ȷ��0.1%���������ֻ������� (����ϸ��ϸ��ϸ�����狀�����Ϊ20%����)��Ʒ��
��3�����������������������������д��������̣���
��4����ʵ������а�������ȫ���գ�����ʵ��������炙��ʵĺ���������ʵ��ֵ����ԭ���� ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2009��ȫ���п���ѧ����������11��ѧ����ʽ������ ���ͣ�������
��09���죩ijͬѧΪ�˼�����ﹺ�������炙����Ƿ�ϸ�����ȡ27.5 g������Ʒ������Ũ�ռ���Һ���ȣ������İ�����100.0g���������ա�������հ�������Һ������m�뷴Ӧʱ��t�ı仯������ͼ��ʾ�����漰�ķ�ӦΪ��
(NH4)2SO4+2NaOH��Na2SO4+2H2O+2NH3���� 2NH3+H2SO4 == (NH4)2SO4 ��
����㣺

��1����ȫ��Ӧ��������� g��
��2���û��ʵĺ�����Ϊ ����ȷ��0.1%����
�����ֻ������� (����ϸ��ϸ�
�ϸ�����狀�����Ϊ20%����)��Ʒ��
��3����������������������������������
���̣���
��4����ʵ������а�������ȫ���գ�����ʵ��������炙��ʵĺ���������ʵ��ֵ����ԭ���� ��
�鿴�𰸺ͽ���>>
����ѧУ��ѡ - ��ϰ���б� - �����б�
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com