
�������ӣ�2005��3��19�գ�ijУ3λѧ����ʵ������в���������ˮ�Ӵ����������֡�������
�Ķ�����������֮��������Ʋ²⣺����������ˮ������Ӧ����ͨ���������ϻ����������Ϣ��
���Ƴ������ڿ������������������ƣ�Na2O���������ƿ���ˮ����������Ӧ�ļ
��������ˮ���ҷ�Ӧ���ų��������ȣ���ѧ����ʽΪ��2Na��2H2O��2NaOH��H2����
������ʦ��ָ������Ʋ�������ʵ�飬����������ʵ���¼��

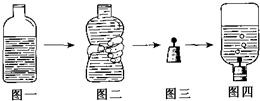
��ȡһ�տ�Ȫˮƿ�����봿��ˮ������һС�ο�϶����ͼһ��ʾ��
�����ּ�ѹ��Ȫˮƿ����ȥ����Ŀ�������ͼ����ʾ��
���ڽ����Ϲ̶�һ����ͷ�룬��������һ������ƣ���ͼ����ʾ��
��Ѹ���ý�����ס�����ƿ�ڣ����ã���ͼ����ʾ��
�ݴ���ȫ��Ӧ��ƿ����������������壻
���ڿ�Ȫˮƿ�е�������A�Լ�����Һ�ʺ�ɫ��
��������Ƶ�ʵ�������ʵ���¼���ش��������⣺
��1��д���Լ�A�����ƣ� ��
��2��д����������ˮ��Ӧ�Ļ�ѧ����ʽ�� ��
��3��Ϊʲô��ʵ�鲽�����Ҫ��ȥ��Ȫˮƿ�еĿ�����
��
��4����ʵ���У���ȡ�õĽ����ƵĴ�С�Ƿ���Ҫ��Ϊʲô��
 �Ķ��쳵ϵ�д�
�Ķ��쳵ϵ�д�
| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ��Ķ�����
 ��2007?���ݣ��������ӣ�2005��3��19�գ�ijУ3λѧ����ʵ������в���������ˮ�Ӵ����������֡�
��2007?���ݣ��������ӣ�2005��3��19�գ�ijУ3λѧ����ʵ������в���������ˮ�Ӵ����������֡��鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��ר���� ���ͣ�ʵ����

�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ������
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��ר��ͻ��ѵ��_������ѧǿ��ϵ��05�������棩 ���ͣ������
�鿴�𰸺ͽ���>>
����ѧУ��ѡ - ��ϰ���б� - �����б�
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com