
| ʵ����� | ÿ�β�����CO2 ��������g�� |
| ��һ�����μ�����100g | |
| �ڶ������μ�����100g | 8.8 |
| �������������100g | 0 |
| 106 |
| x |
| 44 |
| 8.8g |
| 21.2g |
| 25g |
| 106 |
| 21.2g |
| 36.5 |
| 100g��7.3% |
| ʵ����� | ÿ�β�����CO2 ��������g�� |
| ��һ�����μ�����100g | 0 |
| �ڶ������μ�����100g | 8.8 |
| �������������100g | 0 |



| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
 Ϊ�˲ⶨ��ͭ��ͭ��п�Ͻ𣩵���ɣ�ij�о���ѧϰС���ȡ����Ʒ10g����������μ���9.8%��ϡ���ᣬ��Ӧ���������ɵ�����������������Һ��������ϵ��ͼ��ʾ���Լ��㣺
Ϊ�˲ⶨ��ͭ��ͭ��п�Ͻ𣩵���ɣ�ij�о���ѧϰС���ȡ����Ʒ10g����������μ���9.8%��ϡ���ᣬ��Ӧ���������ɵ�����������������Һ��������ϵ��ͼ��ʾ���Լ��㣺�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
| ���� | 1 | 2 | 3 | 4 |
| ����ϡ���������/g | 20.0 | 20.0 | 20.0 | 20.0 |
| ʣ����������/g | 8.7 | 7.4 | 6.1 | 5.8 |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
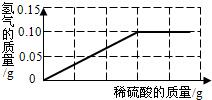
 A��B���ֹ������ʵ��ܽ��������ͼ��ʾ��������������⣺
A��B���ֹ������ʵ��ܽ��������ͼ��ʾ��������������⣺�鿴�𰸺ͽ���>>
����ѧУ��ѡ - ��ϰ���б� - �����б�
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com