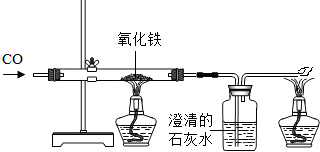
�⣺
��1���������Ͻ�
��2���������������ڿ�����ʱ��������ˮ��Ӧ�Ľ�����ʴ�Ϊ��ˮ��O
2����������
��3����ֹ�������⣬�����ڸ�����ˢ���ᣬ����������ˮ���ʴ�Ϊ���ڱ���ˢ��Ϳ�ͣ�
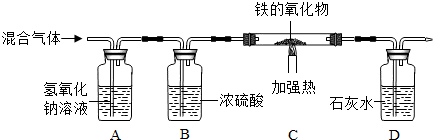
��4����װ��A��ʢ�����������Ƶ�����
�ڵ�Cװ���е����������ﻯѧʽΪFe
XO
Y��ȫ������ԭ��ʣ����������Ϊ14g����֪���ɵ���������Ϊ14g���ֲ��Dװ�õ�����������11g����֪���ɵ�CO
2����Ϊ12g������������Ԫ�ص�һ��������������������������������к�������Ϊ
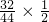
��100%��11g=4g����������ɵã���������������Ԫ�غ���Ԫ�ص�������14g��4g=7��2����ȥ��Aװ�ã��Բⶨ�����Ӱ����Ϊ������̼������ұߵ�װ����ʹ���������ƫС��
��һ����̼�ж���������β������װ�ã�
��5���⣺����������������Ϊx
Fe
2O
3+3CO

2Fe+3CO
2 160 112
x 84t
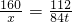
x=120t
�������������120t��90%=133.3t
�������������Ϊ133.3t��
�ʴ�Ϊ����1���ǣ�
��2��ˮ������
��3��ˢ�ᣨ��Ϳ�͵ȣ�
��4���ٳ�������̼��1�֣�����7��2��2�֣���ƫС��1�֣�
���������ռ�β��������D�����ȼ�ŵľƾ��ƻ�ȼ��β������1�֣�
��5���⣺����������������Ϊx
Fe
2O
3+3CO

2Fe+3CO
2 160 112
x 84t
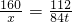
x=120t
�������������120t��90%=133.3t
�������������Ϊ133.3t��
��������1���������Ͻ�
��2��������������������е�������ˮ��Ӧ�Ľ����
��3������ˮ�Ϳ����������ǣ�
��4��ұ��ԭ���IJ��裺�ȳ�������̼���ٸ����ԭ���������������ɵĶ�����̼�ȷ��濼�ǣ�
��5���ȸ��ݻ�ѧ����ʽ�����������Fe
2O
3����������Ȼ���ٸ������������������������Fe
2O
3��90%�ij������������
�����������Ҫ���ջ�ѧ����ʽ����д�����ͽ���������������ֹ��������ķ����ȷ�������ݣ�ֻ���������ܶ���ط��������������ȷ���жϣ�

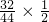 ��100%��11g=4g����������ɵã���������������Ԫ�غ���Ԫ�ص�������14g��4g=7��2����ȥ��Aװ�ã��Բⶨ�����Ӱ����Ϊ������̼������ұߵ�װ����ʹ���������ƫС��
��100%��11g=4g����������ɵã���������������Ԫ�غ���Ԫ�ص�������14g��4g=7��2����ȥ��Aװ�ã��Բⶨ�����Ӱ����Ϊ������̼������ұߵ�װ����ʹ���������ƫС�� 2Fe+3CO2
2Fe+3CO2 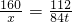 x=120t
x=120t  2Fe+3CO2
2Fe+3CO2 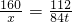 x=120t
x=120t 



 ��������Ҫ�Ľ������ϣ��ڽ��컴����ˮ�ɻ���ʱ�������˴����ĸ�����
��������Ҫ�Ľ������ϣ��ڽ��컴����ˮ�ɻ���ʱ�������˴����ĸ�����