
��������������벻�����ϣ���ÿ��1�֣���10�֣�
��1������ϰ���ϰѽ���������ͭ�������ֽ���ͳ��Ϊ����𡱣��ڡ����˳���аѽ����� ����λ��������һλ�����÷���������ǿ�Ļ��˳��ij�������뵽ϡ��������Һ��Ϊdz��ɫ����Ӧ�Ļ�ѧ����ʽΪ ��
��2��Ϊ�˷�ֹˮ��ͷ���⣬����泣��һ�����������ԭ���ǣ� ������ʱ�����е����Ż���ù��Ǹ�����ԭ����______________________________________��
��3��CO����ұ��ҵԭ�ϣ��������彡���к���д����¯������CO��ԭ�������Ļ�ѧ��Ӧ����ʽ�� ��ú���ж�����ΪCO��ѪҺ�� ��ϣ��������ȱ����
��4������һ��ͭ��п��Ϸ�ĩ���ڿ����г�����պ���ϡ����������ǡ���ܽ⣬�ټ���һ���������ۣ���ַ�Ӧ����ˣ��õ���������Һ����������һ���� ����Һ������һ���� ��
��5�����ÿյ�����ͭ��Ȧ����������ͭ���������� �ԣ�Ҫ��֤þ������ͭ�Ļ��˳����ѡ���˴�ĥ������˿������ͭ��Һ������Ϊ������Ҫ����Һ��______ ______��Һ��

| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
����ÿ����ʴ�����ϵĽ����豸�Ͳ����൱���������20% ~ 40% ��Ϊ̽������ʴ������ijͬѧ��Ƶ�ʵ����ͼ��ʾ��һ�ܺ�۲쵽_______������ţ��Թ��ڵ��������⡣�����������������������ʩ��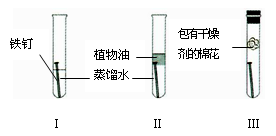
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
��7�֣�ʹ���ܶ�С��ǿ�ȴ��þ�Ͻ��ܼ����������أ��Ӷ������������ĺͷ����ŷš�
|
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
��6�֣����������������ճ������о�����ʹ�á�
��1��������Ͻ������ڸ�ѹ���£���Ҫ�������������� ��
��2�����ڿ����������γ�һ�����ܵ�����Ĥ���Ӷ������������һ���ر����������û�ѧ����ʽ��ʾ������Ĥ���γɹ��� ��
��3�����������ĺϽ��� ����ǡ����ǡ�����
ij�������ó�����ʯ����Ҫ�ɷ�Fe2O3����ԭ��ұ��������Fe2O3��Է�������
Ϊ ��196 t��Fe2O380%�ij����������Ͽ����ú���98�������� t��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
��2�֣���ͬ�����ʣ�����ɡ��ṹ���������ʵȷ��棬�������������������ԡ�������һ�㣬���������Ǹ��õ�ѧ�û�ѧ���������
| �� �� | ������ | ������ |
| Fe��Cu��Mg | ���ܺ�AgNO3��Һ��Ӧ | |
| HCl��H2SO4��HNO3 | | HCl�в�����Ԫ�� |
| C��CO��H2 | | CO�ǻ���������ǵ��� |
| CuSO4��MgSO4��ZnSO4 | ��������ˮ | |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
��9�֣��ճ����������ǹ㷺ʹ�ý������ϡ�
��1�����й��ڽ������ϵ�˵����ȷ���� ������ĸ��ţ���
A�����������������õĽ�������
B��ͭ��Ŀǰ�����������ߵĽ���
C�����������Ԫ������Ȼ�����Ե�����ʽ����
D���ճ�ʹ�õĽ������ϴ�����ǺϽ�
��2��ѡ������Ӳ�ҵIJ��ϲ���Ҫ���ǵ������� ������ĸ��ţ���
A. �����ĵ����� B. ��������ʴ��
C. ������Ӳ�� D. �����۸���Ӳ����ֵ���Ǻ϶�
��3������Ӳ���漰��Fe��Cu��Al��Ni����������֪Ni�ܹ��������·�Ӧ��
Ni+ H2SO4= NiSO4+ H2�� Fe+ NiCl2=" Ni" + FeCl2
��������Ӧ�Ļ������Ͷ��� �����ֽ��������ǿ������˳��Ϊ ����Ԫ�ط��ű�ʾ����
��4����ҵ�������ѳ�����(��Ҫ�ɷ�ΪFe2O3 )�ڸ�¯���CO��Ӧ�����ɵ��������÷�Ӧ�Ļ�ѧ����ʽ�� ��
��������(Fe2O3)����Է��������� ��
��2000t��������84���ij������������Ͽ����ú���98�������� t��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
�ѽӽ����͵��������Һ��Ϊ������Һ�ķ���Ϊ��
�� ��
�ҹ��Ŵ��ͻᣨ��������ͭ��Һ��Ӧ����д���÷�Ӧ�����Ļ�ѧ����ʽ��
�÷�Ӧ�Ļ�����Ӧ����
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
��6�֣������������������о��й㷺��Ӧ�á�
��1�����н�����Ʒ����;�У����ý��������Ե��� ������ĸ��ţ���
��2������ÿ�����Ȼ����ȡ�����Ľ�����������ȡ������������������(��Ҫ�ɷ�ΪFe2O3)��һ����̼�����Ļ�ѧ����ʽΪ ��
��3������Ʒ������ʴ��������������� ��
��4��ij������Һ�к����Ȼ��ơ��Ȼ�ͭ���Ȼ�������ij����С�����÷�Һ��ȡ����ͭ��Cu�������������ֲ�Ʒ����������ͼ��ʾ�������в���������Ԫ�أ���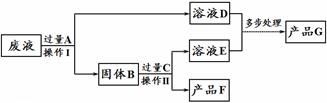
��ش��������⣺
����ҺD�е�����Ϊ ��
�ڼ���C�����ķ����� ��
��A~G���������к���Ԫ�ص������� (����ĸ���)��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
ij����վ���յķϽ�����ĩ�к��̣�Mn������Au����ͭ���ֽ�����Ϊ�˻��չ��ؽ���������վ���й����ֽ���������Ϊ��A��B��C������������»������̣�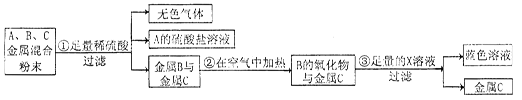
��1��A��B��C���ֽ����Ļ��˳��Ϊ�� �� ��
��2��C�� ��B��������Ļ�ѧʽ�ǣ� ��
��3����֪A����������AԪ����+2�ۣ���д������ٵķ�Ӧ�Ļ�ѧ����ʽ�� ��
��4������Ϊ��������������X��Һ�� ��
��5����ֱ�Ӵ���ɫ��Һ���Ƶý���B����ѡ�õ��Լ��� �����ţ���
a������������Һ b���Ȼ�����Һ c��ϡ���� d����
�鿴�𰸺ͽ���>>
����ѧУ��ѡ - ��ϰ���б� - �����б�
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com