



| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
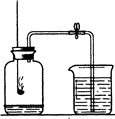 ��2009?����������ͼ��ʾ��ʵ��װ�����ⶨ�����������ĺ�������ش��������⣺
��2009?����������ͼ��ʾ��ʵ��װ�����ⶨ�����������ĺ�������ش��������⣺�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
 ���ڼ䣬�������о�������ɫ����������������ʽ����ѹ����Ȼ��Ϊ���������Ϲ����ŷű��Ĺ�����������Ͷ��ʹ�ã���Ϊ��24����ڼ����ӹ��������ڸ��������ݵ���Ҫ��ͨ���ߣ���ش��������⣺
���ڼ䣬�������о�������ɫ����������������ʽ����ѹ����Ȼ��Ϊ���������Ϲ����ŷű��Ĺ�����������Ͷ��ʹ�ã���Ϊ��24����ڼ����ӹ��������ڸ��������ݵ���Ҫ��ͨ���ߣ���ش��������⣺�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
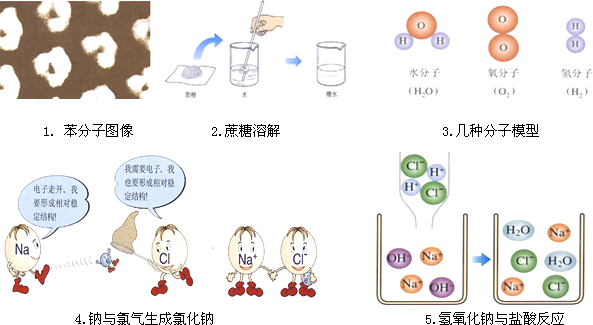
��������������

1. ������ͼ�� 2.�����ܽ� 3.���ַ���ģ��
(1)ͼһ�д��۽Ƕ����õ�һ����Ϣ �� ��
(2)ͼ����ʾ����Ľ��� �� ��
(3)��ͼ����ʾ�������ˮͨ��÷�Ӧ���۹����ǣ���ˮ���ӷֽ�Ϊ��ԭ�Ӻ���ԭ�ӣ��ڱ�ʾΪ �� ���۴����������Ӿۼ�������������������Ӿۼ���������


4.�������������Ȼ��� 5.�������������ᷴӦ
(4)ͼ4��ͨ����˵���ȶ��ṹ��ָ����������� �� ���ӵĽṹ��
(5)����ͼ5�²��������������ᷴӦ����Һ�е������� �� ��
�鿴�𰸺ͽ���>>
����ѧУ��ѡ - ��ϰ���б� - �����б�
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com