��2005?��ijУ��ѧ������ȤС���ͬѧ���о���ѧϰ����չʾ��һ����ͼ��ʾ��ʵ��װ�ã�����ÿ����ѧ��Ӧ����ȫ����������Ʒ�е����ʲ��μӷ�Ӧ����
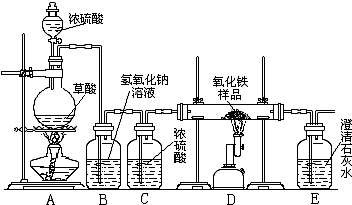
�������ϣ�������Ũ�������ʱ���ȷ������·�Ӧ��
H
2C
2O
4CO��+CO
2��+H
2O
ͨ�����ۣ�ͬѧ�Ƕ�����װ�����˶�����ʶ��
��1����һС��ͬѧ˵����ʵ�鰲ȫ��ʵ�������������Ҫ���װ�õ������ԣ�ʵ�鿪ʼ�ȼ���
A
A
����A��D������ʵ�����ʱ��Ӧ
��
��
�����Ȼ��ֹͣD���ļ��ȣ��ӻ����ĽǶȽ�����Eװ�ú�Ӧ��β�����д������䷽���ǣ�
��ȼ���ռ�����
��ȼ���ռ�����
��
��2���ڶ�С���ͬѧ˵���ø�ʵ��װ�ÿ��Լ���һ����̼����������Ӧ�IJ������Bװ�õ�����
��ȥA�����ɵĶ�����̼
��ȥA�����ɵĶ�����̼
��Eװ�õ�����
���鲢����Dװ�����ɵĶ�����̼
���鲢����Dװ�����ɵĶ�����̼
��һ����̼����������Ӧ�Ļ�ѧ����ʽΪ
��
��3������С���ͬѧ˵����������װ�û����Բⶨ��������Ʒ�����������������������ǵIJⶨ�����ǣ�������������Ʒ������10.0g����Ʒ�벣���ܵ�������Ϊ60.0g����ȫ��Ӧ����ȴ���ٳ�����������ʣ������������Ϊ57.6g������ʵ������������Ʒ������������������Ϊ
80%
80%
��
��4������С��ͬѧ˵����������װ�û�������ⶨ��Ʒ�������������������ķ������ȳ�����������Ʒ���������ٷֱ����Eװ���ڷ�Ӧǰ��������������ɼ��������Ʒ�������������������������˷���ʵ��ʵ��ⶨ���ȴƫ�������ƫ���ԭ�������
Bװ������CO2����֣�E������װ��������е�CO2�������CO2����ĸ���
Bװ������CO2����֣�E������װ��������е�CO2�������CO2����ĸ���
��




