
��ʯ���������ƺ��������ƵĹ��������ʵ���ҳ��õĸ�����������Ϣ����ͼ��ʾ��ͬѧ��Ϊȷ��һƿ���õġ���ʯ�ҡ��������Ѳ��ֱ��ʻ�ȫ�����ʣ���Ʒ�ijɷ֣���������̽����

[�������]ʵ�����о��õļ�ʯ����Ʒ�ijɷ���ʲô��
[���в���] ���õļ�ʯ����Ʒ�п��ܺ���CaO��NaOH��Na2CO3��Ca��OH��2��CaCO3�ȳɷ֡��û�ѧ����ʽ��ʾ��Ʒ�к���Ca��OH��2��Na2CO3���ܺ��е�ԭ�� ��дһ����
[̽������]

�Իش��������⣺
��1�������ٵ������� ����ҺC��һ���� ���ӡ�
��2����ҺA�м���CaCl2��Һ��������壬˵����Ʒ��һ������ ����Ӧ�ķ���ʽΪ ��
��3������ҺA�м���CaCl2��Һ��֤��CaCl2��Һ�����ķ����� ��
[ʵ�����]
��������ʵ��������̼������ۺ϶���Һ����ɷֵ�̽�������ж���Ʒ�ɷֵķ�����ȷ���� ������ţ�
����Ʒ��һ����NaOH ����Ʒ��һ����Na2CO3 ����Ʒ�к�NaOH��CaO�е�һ�ֻ�����
[��չ����]
Ϊ�ⶨʵ������һƿ���ʵ��ռ���NaOH�ĺ�����ijͬѧȡ�������ռ���Ʒ������һ������ˮ�õ�200g��Һ���ټ���200gϡ���ᣨ��������ֽ��赽���ٷų�����Ϊֹ��������ҺΪ395.6g����������㣺
��1����Ӧ������CO2������Ϊ g��
��2������ȡ�ռ���ƷΪ50.0g������Ʒ��NaOH�����������Ƕ��٣���д��������̣�
��3����ͬŨ�ȵ����ᣬ�ֱ���δ���ʡ����ֱ��ʡ���ȫ�����ʣ����ʶ���Na2CO3�����ռ���Ʒ��Ӧ����Ҫ�������������ȡ�����Ԫ�������غ�ĽǶȽ�����ԭ���ǣ� ��
��������[���в���]�����������Ƶķ���ʽΪCaO+H2O�TCa��OH��2 ����̼���Ƶķ���ʽΪ2NaOH+CO2�TNa2CO3+H2O
[̽������]
��1������Һ����IJ���Ϊ���ˣ���̪����ˣ�˵����Һ�Լ��ԣ�����Һ��һ������OH��
��2�������Ȼ��Ʒ�Ӧ����̼���ƣ��䷴Ӧ����ʽΪNa2CO3+CaCl2�T2NaCl+CaCO3��
��3������ȡ��ҺC���Թ��еμ�̼������Һ���а�ɫ�������ɣ���˵��CaCl2��Һ�ѹ���
[ʵ�����]��Ϊ��ˮ���ձ��ڱ���˵����Ʒ�к����������ƻ����������ƣ������Ƕ��߶��У������Ȼ������ɰ�ɫ����˵����Ʒ��һ������̼���ƣ��ۺ����Ϸ�������ѡ�ڢ�
[��չ����]
��1�����������غ㶨�ɿ������Ӧ������CO2������Ϊ200g+200g-395.6g=4.4g
��2�����ݶ�����̼������Ϊ4.4�˾Ϳ��Լ����ˣ����ݻ�ѧ��Ӧ����ʽ���㣬���ɵó���Ʒ��̼���Ƶ��������̶��ó�ԭ��Ʒ���������Ƶ����������岽����𰸡�
��3���������ƺ�̼���ƶ���ÿ46g��Ԫ������142g�����ƣ�����98g����
[���в���]CaO+H2O�TCa��OH��2����2NaOH+CO2�TNa2CO3+H2O�ȣ��𰸺��������֣�
[ʵ�����]��1������ OH��
��2��̼���� Na2CO3+CaCl2�T2NaCl+CaCO3��
��3��ȡ��ҺC���Թ��еμ�̼������Һ���а�ɫ�������ɣ�˵��CaCl2��Һ�ѹ��������������֣�
[ʵ�����]��1���ڢ�
[��չ����]
��1��4.4��������������������������(1��)
��2���⣺����ȡ��Ʒ�к���Na2CO3������Ϊx��
Na2CO3+H2SO4===Na2 SO4+H2O+CO2��������������������������������������(1��)
106 44
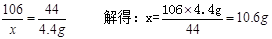 x 4.4g
x 4.4g
��������������������������������(1��)
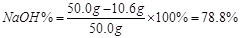
���ռ���Ʒ�� ����������������������(1��)
���ռ���Ʒ��NaOH�İٷֺ���Ϊ78.8%��
��3���������ƺ�̼���ƶ���ÿ46g��Ԫ������142g�����ƣ�����98g���ᡭ��������(1��)


 ���źþ���Ԫ����ĩ��ϵ�д�
���źþ���Ԫ����ĩ��ϵ�д� һ����ʦȨ����ҵ��ϵ�д�
һ����ʦȨ����ҵ��ϵ�д�
| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ��Ķ�����

�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ��Ķ�����

| ||
| ||
| ||
| �� |
| ||
| �� |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
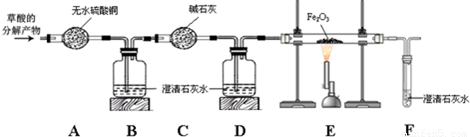
��֪���ᣨH2C2O4������ʱ��Ũ����������»ᷢ���ֽⷴӦ��ij��ѧ��ȤС�����ֽ�������������̽����
��������롿��1���ֽ������һ����̼��ˮ�����Ļ���
��2���ֽ�����Ƕ�����̼��ˮ�����Ļ���
��3���ֽ������һ����̼��������̼��ˮ�����Ļ���
����Ϊ�����IJ����� ��ѡ����ţ��������� ��
���������ϡ���1��Ũ���������ˮ�ԣ����Խ��л����е��⡢��Ԫ�ذ�ˮ�������ȥ��
��2����ɫ����ˮ����ͭ��ˮ���Ϊ��ɫ��
��3����ʯ���ǹ����������ƺ������ƵĻ���
������ʵ�顿Ϊ�˼������ķֽ����ijɷݣ���С��ͬѧ������ķֽ����ͨ����ͼ��ʾ��װ�ã����ּг���������ȥ����

��1��װ��A����ˮ����ͭ������װ��B�г���ʯ��ˮ����ǣ�֤������ֽ������ ����2��װ��D�������� ��
��3��֤������ֽ������һ����̼������Ӧ���� �� ��װ��E�з�����Ӧ�Ļ�ѧ����ʽΪ ��
���ó����ۡ�����ֽ�Ļ�ѧ����ʽΪ ��
����˼�����ۡ��ӻ����Ƕȿ��ǣ�����ʵ��IJ���֮���� ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2011-2012ѧ�꽭��ʡ�������������У�ѧ����У�����꼶���£���ʮ������ĩ��ҵ��ѧ�Ծ��������棩 ���ͣ������

�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2012�꽭��ʡ�Ͼ��а������п���ѧ��ģ�Ծ��������棩 ���ͣ������
�鿴�𰸺ͽ���>>
����ѧУ��ѡ - ��ϰ���б� - �����б�
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com