��2013?�绯����ͼ��ʵ���г�������װ�ã���ش��������⣺
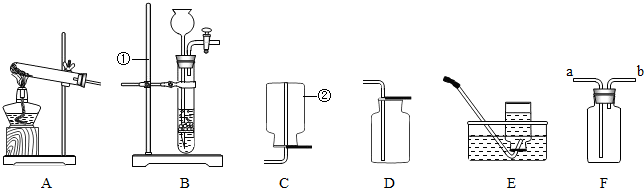
��1��ָ�����б�ŵ��������ƣ�����
����̨
����̨
��
����ƿ
����ƿ
��2��ʵ����������غͶ�����̼��ϼ�����ȡ������Ӧѡ�õķ���װ����
A
A
������ĸ����ͬ�����ռ�װ����
D��E
D��E
������غͶ�����̼��ϼ�����ȡ�����Ļ�ѧ����ʽ��
����ռ����������������ķ�����
�Ѵ����ǵ�ľ�����뼯��ƿ�У���������ǵ�ľ����ȼ��˵���ռ�������������
�Ѵ����ǵ�ľ�����뼯��ƿ�У���������ǵ�ľ����ȼ��˵���ռ�������������
��
��3����ʵ������ȡ������̼���壬Ӧѡ�õķ���װ����
B
B
������ĸ����ͬ�����������̼�����ռ����ķ�����
��ȼ�յ�ľ�����ڼ���ƿ�ڣ���������ǵ�ľ��Ϩ��˵���Ѿ��ռ���
��ȼ�յ�ľ�����ڼ���ƿ�ڣ���������ǵ�ľ��Ϩ��˵���Ѿ��ռ���
��
��4����ͼ��Fװ���ж�����;������Fװ���ռ�����������Ӧ��
b
b
���a����b�����˵��ܵ��룮����Fװ�ó�ȥһ����̼�����л��еĶ�����̼���壬ƿ��Ӧʢ�ŵ��Լ���
NaOH
NaOH
��Һ���û�ѧʽ��д������Ӧ�Ļ�ѧ����ʽΪ
2NaOH+CO2�TNa2CO3+H2O
2NaOH+CO2�TNa2CO3+H2O
����5����������̼����ͨ����ɫʯ����Һ�У����Կ�����ɫʯ����Һ��
��
��
ɫ��ԭ����
CO2+H2O=H2CO3
CO2+H2O=H2CO3
���û�ѧ����ʽ��ʾ����





 ��2013?�绯����ͼ��ij��ѧ��Ӧǰ����۹���ʾ��ͼ�����С�
��2013?�绯����ͼ��ij��ѧ��Ӧǰ����۹���ʾ��ͼ�����С� ������
������ ����ʾ���ֲ�ͬ��ԭ�ӣ������ͼʾ�ش�
����ʾ���ֲ�ͬ��ԭ�ӣ������ͼʾ�ش� ��
�� ��
�� �������ӵĸ�������
�������ӵĸ������� ��2013?�绯����ͼ��Ԫ�����ڱ�����Ԫ�ص������Ϣ��д��������ͼ�л�õ���Ϣ��
��2013?�绯����ͼ��Ԫ�����ڱ�����Ԫ�ص������Ϣ��д��������ͼ�л�õ���Ϣ��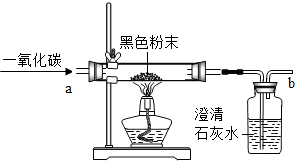 ��2013?�绯����̽�����ϣ���ʦ��ͬѧ��һ����ɫ��ĩ���÷�ĩ�������ۺ�����ͭ��ĩ�е�һ�ֻ�������ɣ�Ϊ��ȷ���÷�ĩ�ijɷ֣�ͬѧ�ǽ�������̽����
��2013?�绯����̽�����ϣ���ʦ��ͬѧ��һ����ɫ��ĩ���÷�ĩ�������ۺ�����ͭ��ĩ�е�һ�ֻ�������ɣ�Ϊ��ȷ���÷�ĩ�ijɷ֣�ͬѧ�ǽ�������̽���� ��2013?�绯����ѧϰ������ȼ�յ�̽��ʵ���׳׳ͬѧ�ص������ֽ�����һ��ʵ�����
��2013?�绯����ѧϰ������ȼ�յ�̽��ʵ���׳׳ͬѧ�ص������ֽ�����һ��ʵ�����