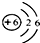̼���γɻ�������������Ԫ�أ���̼��������ѧ��ѧ�о�����Ҫ���ݣ�
��1����ͼΪԪ�����ڱ��е�һ������˵������ȷ����______�����ţ���
A��̼Ԫ�����ڷǽ���Ԫ��
B��̼ԭ�Ӻ���������Ϊ6
C��̼Ԫ�ص�ԭ�ӽṹʾʾ��ͼΪ
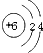
D��̼�����ԭ������Ϊ12.01g
��2��ʵ�����У���������װ�ÿ�����ȡ���壬��ش��������⣮
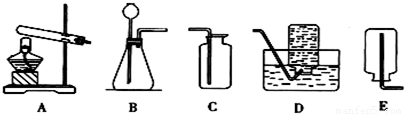
���ô���ʯ��ϡ���ᷴӦ��ȡCO
2��Ӧѡ�õķ���װ���ǣ�����ţ���ͬ��______�����ѡ�õ��ռ�װ����______��
�ڼ��ȹ���̼�����ƺ���̼����臨��ܲ���CO
2���仯ѧ����ʽ�ֱ��ǣ�
2NaHCO
3
Na
2CO
3+H
2O+CO
2�� NH
4HCO
3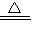
NH
3��+H
2O+CO
2��
ijͬѧ���ü��ȹ���̼�����Ƶķ�����ȡCO
2��Ӧ��ѡ�õķ���װ����______������ѡ����ȹ���̼�������ȡCO
2��������______��
��Aװ�����Թܿ���������б��ԭ����______��
��3����֪������̼����ˮ��Ӧ�����ᣮijͬѧ���������ʵ�飬��֤����һ���ۣ�
ʵ��1��ȡ4����ʯ����ҺȾ����ɫ�ĸ����ֽ��������һ��ֽ��������ϡ���ᣬС�����ɫ��
ʵ��2�����ڶ���ֽ��������ˮ��ֽ������ɫ��
ʵ��3��
ʵ��4�������Ķ�ֽ������ˮ���ٷ���ʢ��CO
2�ļ���ƿ�У�ֽ�����ɫ��
�ش��������⣺
��ʵ��2��Ŀ����______��
���벹��ʵ��3��______��
�۶�����̼��ˮ��Ӧ�Ļ�ѧ����ʽΪ______��
��4����ȡ12.5gʯ��ʯ����Ҫ�ɷ���CaCO
3�����ʲ��μӷ�Ӧ�������ձ��У������м���50gϡ���ᣨ����û�������꣩����Ӧ����������ձ���ʣ�����ʵ�������Ϊ58.1g���������ձ������������Լ���ʯ��ʯ�����ʵ�����������




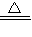 Na2CO3+H2O+CO2�� NH4HCO3
Na2CO3+H2O+CO2�� NH4HCO3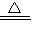 NH3��+H2O+CO2��
NH3��+H2O+CO2��
 ���X=10g�����Ժ�����12.5g-10g=2.5g��������������������
���X=10g�����Ժ�����12.5g-10g=2.5g�������������������� ×100%=25%��
×100%=25%��


 ̼���γɻ�������������Ԫ�أ���̼��������ѧ��ѧ�о�����Ҫ���ݣ�
̼���γɻ�������������Ԫ�أ���̼��������ѧ��ѧ�о�����Ҫ���ݣ� ̼���γɻ�������������Ԫ�أ���̼��������ѧ��ѧ�о�����Ҫ���ݣ�
̼���γɻ�������������Ԫ�أ���̼��������ѧ��ѧ�о�����Ҫ���ݣ�
 ̼���γɻ�������������Ԫ�أ���̼��������ѧ��ѧ�о�����Ҫ���ݣ�
̼���γɻ�������������Ԫ�أ���̼��������ѧ��ѧ�о�����Ҫ���ݣ� D��̼�����ԭ������Ϊ12.01
D��̼�����ԭ������Ϊ12.01
 ̼���γɻ�������������Ԫ�أ���̼��������ѧ��ѧ�о�����Ҫ���ݣ�
̼���γɻ�������������Ԫ�أ���̼��������ѧ��ѧ�о�����Ҫ���ݣ�