
| 32��1 |
| 32��1+16��2 |
| 64 |
| 254 |
| X |
| 2g��0.0254% |


 ������ÿ�ʱ�Ż���ҵϵ�д�
������ÿ�ʱ�Ż���ҵϵ�д�
| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ��ʴ���
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��ͬ���� ���ͣ������
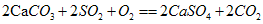 ��ʵ��֤��ʯ�ҽ�[Ca(OH)2]�ڿ�����Ҳ������SO2��������ƺ��������ʣ�д���÷�Ӧ�Ļ�ѧ����ʽ��________________________________��
��ʵ��֤��ʯ�ҽ�[Ca(OH)2]�ڿ�����Ҳ������SO2��������ƺ��������ʣ�д���÷�Ӧ�Ļ�ѧ����ʽ��________________________________���鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2003���ʮ���조��ԭ����ȫ������ѧ����ѧ���ʺ�ʵ���������������������������Ծ��������棩 ���ͣ������
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ����7.3 ʹ��ȼ�϶Ի�����Ӱ�졷2010��ͬ����ϰ��1���������棩 ���ͣ������
�鿴�𰸺ͽ���>>
����ѧУ��ѡ - ��ϰ���б� - �����б�
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com