
��9�֣���������ۺ����ۡ�����ۡ��۸����ܴ���ۡ��ָ�û�����Բ��죬ij��ѧ��ȤС��ͬѧ�Զ��߳ɷֵIJ�ͬ������̽����
���������ϡ�
�����������������ӹ����ɣ�����һ������̼��ơ�������ȣ�����������ܽ���ˮ������Ũ���Ტ���Ȼ���ֻ�ɫ���ɫ��
�����ۡ�����ۡ��ɱ��Ǽӹ����ɣ��ӹ������л��õ��������ƣ����ǵ���Ҫ�ɷ���̼��ơ�
����������ۺ����ۡ�����ۡ��У���̼����⣬�����ɷ־����������ᷴӦ���ɶ�����̼��
��������롿��������ۺ����ۡ�����ۡ�����������У�
�����ۡ�����ۡ��к��� ��
�����ۡ�����ۡ��в��������ᡣ
����������ۺ����ۡ�����ۡ���̼��Ƶ�����������ͬ��
��ʵ���������������ۺ����ۡ�����ۡ��ֱ������ֻ�ձ��У�������ˮ�ܽ⣬����һ��ʱ�����ˣ�ȡ������Һ���á�
| ���� | ���� | ���� |
| ȡ����������Һ�ֱ�����֧�Թ��У��μ����� | ��������۵���Һû�б�ɫ�����ۡ�����ۡ�����Һ��� | ����ٳ��� |
| ȡ����������Һ�ֱ�����֧�Թ��У�����һ���� ������������ | ��������۵���Һ�л�ɫ���֣��ֲ���ڣ����ۡ�����ۡ�����Һû���������� | ����ڳ��� |
| | ��������� | ���ۡ�����ۡ� |
| �����ĩ������ | 10.00 g | 10.00 g |
| ����ϡ��������� | 46.00 g | 50.13 g |
| ��Ӧ���ձ������ʵ����� | 52.00 g | 55.77 g |
����������������ۡ�����ۡ��ɱ��Ǽӹ����ɣ��ӹ������л��õ��������ƣ��������ۡ�����ۡ��к����������ƣ����ۡ�����ۡ��к����������ƣ��������Ƴʼ��ԣ�����Ӧ������Һ�м����̪��Һ����Һ�ʼ��ԣ���̪���죬��������۵���Һ���а����ᣬ���������Ե����ʣ�����Ӧ�������ᣬijЩ�����������������ɫ���ɼ���ɵó����ۡ�����ۡ���̼��Ƶ���������������������ۣ�������������л����������������ƣ����Լ����ϡ������Ⱥ��������Ʒ�Ӧ�����¼���Ľ������
���㣺ʵ��̽����



| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ�̽����
ijУ��ѧ��ȤС��ͬѧ�������غ㶨�ɽ���ʵ��̽��������ͬѧ���ڿ�����ȼ��þ����ʵ����̽��������ͬѧ��ͼ������̽�����ش��������⣺
��1������ͬѧ��þ��ȼ�պ�������ʯ�����Ϲ�������������ֱȷ�Ӧǰþ��������Ҫ�ᡣ����������е�ԭ������� ��������������ͬѧ��ȼ�յ�þ���Ϸ������֣�ʹ�гֵ�þ����ȫȼ�ղ�ʹ������ȫ���ռ�������������Ƶõ�������ԭþ�������� �����ȡ����С�������������ԭ�� ��
��2��̼���������ᷴӦ�Ļ�ѧ����ʽΪ ������ͬѧʵ�������ǣ���ƽ �������ƽ�⡱������ƫת��������ƫת����
��3��ͨ����������ʵ�飬���õ���ʾ�� ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�̽����
��11�֣������װ�г�ʹ��һ�ִ�װ��������Ʒ��Ϊ��504˫�����������ǩ����ͼ��ʾ��ͬѧ�Ƕ�һ�����õġ�504˫������������Ʒ�ܺ��棬���ʵ�����̽����
��������⡿���ù���ijɷ���ʲô��
���������ϡ������Ȼ�����Һ�ڳ����·�����Ӧ�����Ȼ�������
���������롿���ù����п��ܺ���Fe��Fe2O3��CaO��Ca(OH)2��CaCO3��
���ù����п��ܺ���Ca(OH)2��ԭ����(�û�ѧ����ʽ��ʾ) ��
��ʵ��̽��1��
��ͬѧ�ķ�����
| ʵ����� | ʵ������ | ʵ����� |
| ��ȡ������������Թ��У�����������ˮ�ܽ⣬���ú�ȡ�ϲ���Һ�μ���ɫ��̪��Һ�� | �����ܽ�ʱ�Թ���ڷ��̣��Թܵײ��в������Һ��졣 | ������һ������ �� ���������ơ� |
| ����ȡ������������Թ��У��μ�����ϡ���ᡣ | ��������ʧ���д�����ɫ����������õ�dz��ɫ��Һ�� | ������һ������ �� �� һ������Fe2O3�� |
| �ǽ�������в���������ͨ�뵽�����ʯ��ˮ�С� | �� | ������һ������CaCO3�� |

�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�̽����
��7�֣���һ����ɫ������Ʒ��������̼���ơ����ᱵ���������ơ��Ȼ����е�һ�ֻ��֡�Ϊ̽����ɷ֣�С����ʦȡ��һ������Ʒ��������ˮ�ܽ⣬���˵õ���ɫ��������ɫ��Һ�ҡ�
д�����������п��ܷ�����Ӧ�Ļ�ѧ����ʽ�� ��
����ʱ�õ����������������������� ��
��̽���һ�� �ܽ�С��̽����ɫ�����ijɷ֡�
| ʵ����� | ʵ������ | �� �� |
| ȡ��ɫ�����ף��μ�������ϡ���� | ����������ʧ | ��ɫ��������һ������ |
| ʵ����� | ʵ������ | ���� | |
| ʵ��� | ȡ������ɫ��Һ�ң�����̼������Һ | ���������� | ��ɫ��Һ����һ������ |
| ʵ��� | ����٣�ȡ������ɫ��Һ�ң���������� ��Һ������ | ���ɰ�ɫ���� | ԭ��ɫ������Ʒ��һ�����ڸ����� |
| ����ڣ�ȡ������е���Һ���μ� ��Һ | | ||
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�̽����
��18�֣���ҵ�ռNaOH�����нϺõ�ɱ�����������������ã�����ҵ�ռ��г���������̼���ơ�ij��ѧѧϰС��ͬѧΧ�ƹ�ҵ�ռ�չ��ϵ���о���
̽��һ����ҵ�ռ����Ƿ���̼����
���������ϡ�̼���ƺ��Ȼ����ܷ������ֽⷴӦ��
С��ָ����ѡ������ʵ��ҩƷ��̽����ϡ���ᡢ��̪��Һ��CaCl2��Һ��
��1��С��ͬѧ�������ۺ�һ����Ϊʹ�� ҩƷ���ܴﵽ̽��Ŀ�ģ������� ��
��2�������ѡ�õ�ʵ��ҩƷ�������ʵ�鱨�棺
| ʵ����� | ʵ������ | ʵ����� |
| ȡ������ҵ�ռ���Ʒ�����Һ�μӹ��� | | ��ҵ�ռ��к���̼���ƣ�����ʵ������Ļ�ѧ����ʽ�� �� |

�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�̽����
��8�֣���1��С������ͼ��ʾװ�öԶ�����̼�����ʵ������о���
�۲쵽��������____________________________________��
ʵ��Ľ�����______________________________________��
��2����������⡿������̼�ܷ�֧��ȼ�գ�
��������衿þ���ڶ�����̼��ȼ�ա�
���������ϡ�������þ��������þ���ǰ�ɫ������ˮ�Ĺ��塣
��MgO+2HCl= MgCl2+H2O ��
��MgCl2+2NaOH=Mg(OH)2��+2NaCl ��
��ʵ��̽����
����ͼ��þ������ȼ�գ�ð���̣��к�ɫ�������ɣ����ų��������ȡ�
��Ϊ����������ijɷ֣�������ʵ�顣
| ʵ�鲽�� | ʵ������ | ʵ����ۺͻ�ѧ����ʽ | |
| ����ƿ�м���������ᣬ��ַ�Ӧ����ˣ�����ֽ�����к�ɫ���塣 | I������ɫ�����ռ���ϴ�ӡ������ȼ���ڻ����Ϸ���һ��պ�г���ʯ��ˮ���ձ��� | ��ɫ����ȼ�գ��ձ��ڱڳ��ְ�ɫ���ǡ� | ��ɫ������______�� ��Ӧ�Ļ�ѧ����ʽ�ǣ� ___________________�� |
| II��ȡ������Һ���Թ��У���μ�������������Һ�� | ��ʼ_______�����а�ɫ���������� | ���̵ijɷ���______�� | |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�̽����
��6�֣���һʵ����ʵ������������ͭ��Һ��Ӧ��������ͭ���ɣ������н϶�����������Ϊȷ������ijɷ֣���������ʵ��̽����
��һ��̽�������������Ԫ�ؽǶȷ��������������SO2��O2��H2�е�һ�ֻ��֡�
������ʵ�飺
��1����ͬѧΪȷ���Ƿ���SO2����������ͨ�����Ը��������Һ�У���Һ��ɫδ�����仯����������� SO2����С����ޡ�����
��2����ͬѧ��Ϊֻ��O2������� �����顣ʵ��֤����ͬѧ�Ĺ۵㲻��ȷ
��3����ͬѧΪ�ж��Ƿ���O2��ͬʱȷ������ɷ֣�ͨ���������ϵ�֪��
O2+4KI+4HCl ="==" 2I2+4KCl+2H2O��I2Ϊ�ⵥ�ʡ���������������µ�ʵ��װ�ã�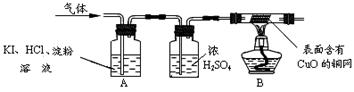
������ʵ����������ۣ�A�й۲쵽 ��֤����O2��
B�й۲쵽 ��֤����������H2��
���ģ�ʵ�鷴˼��
��4�������Ϊ��ͬѧ�ڼ���֮ǰ��Ӧ�������� ��ȷ����ȫ��
��5��������ʵ���֪������ͭ��Һ�� �ԣ���ᡱ�����
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�̽����
��ʵ��̨������ƿδ����ǩ����Һ����֪�ֱ���̼������Һ������������Һ��ϡ���ᡣΪ������������Һ������ʦָ���£���ȤС���ͬѧ����������Һ��A��B��C���б�ţ�Ȼ��ֱ��ȡ������Ϊ��Ʒ���뵽��֧�Թ��У�����������ͼ��ʾ��̽����� 
����һ�У�C����������A��B��Һ����ɫ��ɺ�ɫ����A�к�ɫ������ȥ�ָ�Ϊ��ɫ��
������У�B��Һ����ɫǡ���ɺ�ɫ�����ɫ������Һ��������ð����A��Һ�ֱ�Ϊ��ɫ��
���й��ڴ�ʵ�����������ȷ����
A�� C��Һ������������Һ
B�� B��Һ��ϡ����
C�� �������ʵ���B�Թ����������Ȼ��ơ���̪
D�� �������ʵ���A�Թ����������Ȼ��ơ���̪
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�̽����
Ϊ��֤���������H2��CO��HC1��ɣ������ͼ��ѡ���ʵ���ʵ��װ����
��һ��ʵ�飬���ش���������
��1������ѡ��װ�õ���ȷ˳����(ֻҪ��д��ͼ��װ�õ����)
��2������װ�õ�����˳����(����ӿڵ���ĸ�����ӽ�ʡ�ԣ�װ��֮���á���������)�������� ��
��3������ѡ�õ�װ�õ����� ���ڶ���ѡ�õ�װ�õ����� ��
��4��������������� ��
�鿴�𰸺ͽ���>>
����ѧУ��ѡ - ��ϰ���б� - �����б�
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com