
 ʵ������С���߽�ʵ���Һ���ʦһ����ÿ��ʵ�����ϵ�ҩƷ�������Ƿ��뱸���ߵ�ij���ʱ������һ������г�ġ�����������ͼ����
ʵ������С���߽�ʵ���Һ���ʦһ����ÿ��ʵ�����ϵ�ҩƷ�������Ƿ��뱸���ߵ�ij���ʱ������һ������г�ġ�����������ͼ����| ̽��Ŀ�� | ̽������ | Ԥ������ |
| ������Һ�е�CO32- | �٣�ȡ������Һ���Թ��У��μ������� �Ȼ��� �Ȼ��� �Լ� |
�а�ɫ�������� |
| ֤����Һ���д�NaOH | �ڣ���ʵ���������Һ�еμӷ�̪��Һ | ��̪��� ��̪��� |


 ��ҵ����ϵ�д�
��ҵ����ϵ�д� ͬ��ѧ��һ�ζ���ϵ�д�
ͬ��ѧ��һ�ζ���ϵ�д� �����ܾ�ϵ�д�
�����ܾ�ϵ�д� ���ƿ�����ϵ�д�
���ƿ�����ϵ�д�
| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
 24��һ�죬ʵ������С���С���߽�ʵ���ң�����ʦһ����ÿ��ʵ�����ϵ�ҩƷ�������Ƿ��뱸���ߵ�ij���ʱ������һ������г�ġ�����������ͼ����
24��һ�죬ʵ������С���С���߽�ʵ���ң�����ʦһ����ÿ��ʵ�����ϵ�ҩƷ�������Ƿ��뱸���ߵ�ij���ʱ������һ������г�ġ�����������ͼ�����鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�

| ̽��Ŀ�� | ̽������ | Ԥ������ |
| ������Һ�е�CO32- | ��ȡ������Һ���Թ��У��μ��������Լ� | ������ |
| ֤����Һ���д�NaOH | ����ʵ���������Һ�еμӷ�̪��Һ |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
 26��һ�죬ʵ������С���߽�ʵ���ң�����ʦһ����ÿ��ʵ�����ϵ�ҩƷ�������Ƿ��뱸���ߵ�ij���ʱ������һ������г�ġ�������������ͼ����
26��һ�죬ʵ������С���߽�ʵ���ң�����ʦһ����ÿ��ʵ�����ϵ�ҩƷ�������Ƿ��뱸���ߵ�ij���ʱ������һ������г�ġ�������������ͼ����
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
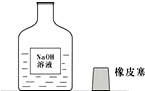 һ�죬ʵ������С���߽�ʵ���ң�����ʦһ����ÿ��ʵ�����ϵ�ҩƷ�������Ƿ��뱸���ߵ�ij���ʱ������һ������г�ġ�����������ͼ��
һ�죬ʵ������С���߽�ʵ���ң�����ʦһ����ÿ��ʵ�����ϵ�ҩƷ�������Ƿ��뱸���ߵ�ij���ʱ������һ������г�ġ�����������ͼ��
| ||
. |
| ̽��Ŀ�� | ̽������ | Ԥ������ |
| ������Һ�е�CO32- | aȡ������Һ���Թ��У��μ������� |
|
| ֤����Һ���д�NaOH | b��ʵ��a������Һ�еμӷ�̪��Һ |
�鿴�𰸺ͽ���>>
����ѧУ��ѡ - ��ϰ���б� - �����б�
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com