
��ǰ��������۵ļ۸����Ԫ���ϰ�Ԫ���ȣ�������ۡ��ָж�û�����Բ��졣
��ô��������ۺ���������۵ijɷ��кβ�ͬ�أ��ʹ����⣬ʵ��С��ͬѧչ��̽����
���������ϡ������������ֱ���ɱ��ƶ�����������ӹ����ɣ�����һ�����İ����ᡢ̼��Ƶȡ�
������������ɡ����Ƿۡ��ӹ����ɣ����Ǽӹ������л��õ��������ƣ����ǵ���Ҫ�ɷ���̼��ơ�
��������г�̼����⣬�����ɷ־����������ᷴӦ���ɶ�����̼��
�ܰ���������ܽ���ˮ������Ũ���Ტ���Ȼ���ֻ�ɫ���ɫ��
���Ȼ�����Һ�����ԡ�
��������롿��������ۺ���������۵�����������ڣ�����������ۺ��� ������������۲����������̼��ƺ�����ͬ��
��ʵ��̽����
|
ʵ������ |
���� |
���� |
|
��1���ֱ�������������ۺ���������۷����Թ��У�������ˮ������һ��ʱ��μ� �� |
��������۵��ϲ���Һ��죬��������۵���Һû�б�ɫ |
����ٳ��� |
|
��2���ֱ�������������ۺ���������۷����Թ��У�������ˮ�����ˣ� �����ȡ� |
��������۵���Һ�л�ɫ���֣��ֲ���ڣ���������۵���Һû���������� |
���� ���� |
��3���ֱ�һ����������������ۺ���������۷����ձ��У�����μ���14.6%��ϡ���ᣬ�ߵμӱ���ֱ��ǡ����ȫ��Ӧ�����ݼ�¼���£�
|
|
��������� |
��������� |
|
����۵����� |
100 g |
100 g |
|
������������� |
460.0 g |
500.0 g |
|
�ձ����������ʵ������� |
520.0 g |
558.2 g |
�ٴ�������ʱ��С�����������˼·���ü������������������������������������Ӧ�������е������������������û�ѧ����ʽ����������̼��Ƶ�������
����ΪС����˼·�Ƿ���У������� ��
�ڴ������ݺó����ۣ������������̼��Ƶ���������Ϊ %������������������ۡ�
��������˼��ͨ��̽���ͽ�����ͬѧ����Ϊ������������ۺ���������۵��������еķ����� ��
��������롿NaOH ��ʵ��̽������1����̪��Һ ��2������Һ�м���Ũ���� ��
��3�������У���Ϊ������л���NaOH������Ҳ�������ᷴӦ 95.0
��������˼��ȡ������Ʒ�����Թ��У�������ˮ������һ��ʱ��μӷ�̪��Һ�����Թ����ϲ���Һ��죬����ƷΪ��������ۣ�����������������ƷΪ��������ۡ�
��������
�����������������롿�ٸ�����������۵ļӹ����̿�֪�������ڼӹ�������Ҫʹ���������ƣ�������������к����������ơ�
��ʵ��̽������1�����ʵ����ۺ�ʵ�������������������������к����������ƣ�����Һ�ʼ��ԣ�������ʹ��̪��Һ��죬�ʵμӵ��Ƿ�̪��Һ��
��2������ʵ��������������۵���Һ�л�ɫ���֣��ֲ���ڣ���������۵���Һû�������������������Ͽ�֪������Һ�м������Ũ���ᣬ��Ϊ����������Ũ���Ტ���Ȼ���ֻ�ɫ���ɫ���ɴ˿�֪������ڳ�����
��3���ٲ����У���������������к��е��������ƻ������ᷢ����Ӧ���ʲ���ֱ�Ӽ���ó������
�ڸ��ݱ����е����ݿ�֪����������������ᷴӦ�������Ķ�����̼������=100g+500g-558.2g=41.8g
������������к�̼��Ƶ�����Ϊx
CaCO3+2HCl=CaCl2+H2O+CO2��
100 44
x 41.8g
100��x=44��41.8g
��ã�x=95g
������������к�̼��Ƶ���������=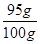 ��100%=95%
��100%=95%
��������˼�����ݶ��ߺ��еIJ�ͬ�����ʵ����ʵIJ�����з��������������Ƶ�ˮ��Һ�ʼ��ԣ���ʹ��̪��Һ��죬�ʿ�ȡ������Ʒ�����Թ��У���������ˮ������һ��ʱ��μӷ�̪��Һ�����Թ����ϲ���Һ��죬����ƷΪ��������ۣ�����������������ƷΪ��������ۡ�
���㣺ʵ��̽�����ʵ���ɳɷ��Լ�����
������������֤��ʵ��̽����Ҫ������ʵ����ʻ�仯���ɣ����ݸ�����ʵ����Ʒ���������ʵ�顢������̽������ͨ���۲졢��¼�ͷ�����ʵ����������֤�����ʵ����ʻ�仯���ɵȡ����о��������ʱ��ͨ�����Ե�̽��ʵ�鲻��ȷ��ij��ɳɷ�ʱ����ͨ�������о������жϣ������о�ʱ�������غ�˼���ǽ����ƶϵĸ������ݡ�



| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ��Ķ�����
 ��2013?������һģ����ǰ��������۵ļ۸����Ԫ���ϰ�Ԫ���ȣ�������ۡ��ָж�û�����Բ��죮
��2013?������һģ����ǰ��������۵ļ۸����Ԫ���ϰ�Ԫ���ȣ�������ۡ��ָж�û�����Բ��죮| ʵ������ | ���� | ���� |
| ��1���ֱ�������������ۺ���������۷����Թ��У�������ˮ������һ��ʱ��μ� ��̪��Һ ��̪��Һ �� |
��������۵��ϲ���Һ��죬��������۵���Һû�б�ɫ | ����ٳ��� |
| ��2���ֱ�������������ۺ���������۷����Թ��У�������ˮ�����ȣ� | ��������۵���Һ�л�ɫ���֣��ֲ���ڣ���������۵���Һû���������� | ����ڳ��� |
| ��������� | ��������� | |
| ����۵����� | 100 g | 100 g |
| ������������� | 460.0 g | 501.3 g |
| �ձ����������ʵ������� | 520.0 g | 557.7 g |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ��Ķ�����
| ʵ������ | ���� | ���� |
| ��1���ֱ�������������ۺ���������۷����Թ��У�������ˮ������һ��ʱ��μ� ��̪��Һ ��̪��Һ �� |
��������۵��ϲ���Һ��죬��������۵���Һû�б�ɫ | ����ٳ��� |
| ��2���ֱ�������������ۺ���������۷����Թ��У�������ˮ�����ˣ� ����Һ�м���Ũ���� ����Һ�м���Ũ���� �����ȣ� |
��������۵���Һ�л�ɫ���֣��ֲ���ڣ���������۵���Һû���������� | ���� �� �� ���� |
| ��������� | ��������� | |
| ����۵����� | 100g | 100g |
| ������������� | 460.0g | 500.0g |
| �ձ����������ʵ������� | 520.0g | 558.2g |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2013�������������������б�ҵ����Ĵν�ѧ����������⻯ѧ�Ծ����������� ���ͣ�̽����
��ǰ��������۵ļ۸����Ԫ���ϰ�Ԫ���ȣ�������ۡ��ָж�û�����Բ��졣
��ô��������ۺ���������۵ijɷ��кβ�ͬ�أ��ʹ����⣬ʵ��С��ͬѧչ��̽����
���������ϡ������������ֱ���ɱ��ƶ�����������ӹ����ɣ�����һ�����İ����ᡢ̼��Ƶȡ�
������������ɡ����Ƿۡ��ӹ����ɣ����Ǽӹ������л��õ��������ƣ����ǵ���Ҫ�ɷ���̼��ơ�
��������г�̼����⣬�����ɷ־����������ᷴӦ���ɶ�����̼��
�ܰ���������ܽ���ˮ������Ũ���Ტ���Ȼ���ֻ�ɫ���ɫ��
���Ȼ�����Һ�����ԡ�
��������롿��������ۺ���������۵�����������ڣ�����������ۺ��� ������������۲����������̼��ƺ�����ͬ��
��ʵ��̽����
| ʵ������ | ���� | ���� |
| ��1���ֱ�������������ۺ���������۷����Թ��У�������ˮ������һ��ʱ��μ� �� | ��������۵��ϲ���Һ��죬��������۵���Һû�б�ɫ | ����ٳ��� |
| ��2���ֱ�������������ۺ���������۷����Թ��У�������ˮ�����ˣ� �����ȡ� | ��������۵���Һ�л�ɫ���֣��ֲ���ڣ���������۵���Һû���������� | ���� ���� |
| | ��������� | ��������� |
| ����۵����� | 100 g | 100 g |
| ������������� | 460.0 g | 500.0 g |
| �ձ����������ʵ������� | 520.0 g | 558.2 g |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2012-2013ѧ�걱���к������п�һģ��ѧ�Ծ��������棩 ���ͣ�̽����
��ǰ��������۵ļ۸����Ԫ���ϰ�Ԫ���ȣ�������ۡ��ָж�û�����Բ��죮
��ô��������ۺ���������۵ijɷ��кβ�ͬ�أ��ʹ����⣬ʵ��С��ͬѧչ��̽����
���������ϡ�
�����������ֱ���ɱ��ƶ�����������ӹ����ɣ�����һ�����İ����ᡢ̼��Ƶȣ�
������������ɡ����Ƿۡ��ӹ����ɣ����Ǽӹ������л��õ��������ƣ����ǵ���Ҫ�ɷ���̼��ƣ�
��������г�̼����⣬�����ɷ־����������ᷴӦ���ɶ�����̼��
�ܰ���������ܽ���ˮ������Ũ���Ტ���Ȼ���ֻ�ɫ���ɫ��
��̼�����Ũ�����ܷ�����Ӧ�����ɶ�����̼��
���Ȼ�����Һ�����ԣ�
��������롿
��������ۺ���������۵�����������ڣ�
����������ۺ����� ��
����������۲��������
��̼��ƺ�����ͬ��
��ʵ��̽����
|
ʵ������ |
���� |
���� |
|
��1���ֱ�������������ۺ���������۷����Թ��У�������ˮ������һ��ʱ��μ��� ���� |
��������۵��ϲ���Һ��죬��������۵���Һû�б�ɫ |
����ٳ��� |
|
��2���ֱ�������������ۺ���������۷����Թ��У�������ˮ�����ȣ� |
��������۵���Һ�л�ɫ���֣��ֲ���ڣ���������۵���Һû���������� |
����ڳ��� |
��3���ֱ�һ����������������ۺ���������۷����ձ��У�����μ���14.6%��ϡ���ᣬ�ߵμӱ���ֱ��ǡ����ȫ��Ӧ�����ݼ�¼���£�
|
|
��������� |
��������� |
|
����۵����� |
100 g |
100 g |
|
������������� |
460.0 g |
501.3 g |
|
�ձ����������ʵ������� |
520.0 g |
557.7 g |
�ٴ�������ʱ��С�����������˼·���ü������������������������������������Ӧ�������е������������������û�ѧ����ʽ����������̼��Ƶ�����������ΪС����˼·�Ƿ���У��������� �ڴ������ݺó����ۣ������������̼��Ƶ���������Ϊ����%������������С�����1λ��������������������ۣ�
��ʵ�鷴˼��
������˼��ͬѧ����Ϊ���������Լ���ֻ���Ե����������Ϳ���ͨ��һ������۵�ȡ����������������֤����д������������� ��

�鿴�𰸺ͽ���>>
����ѧУ��ѡ - ��ϰ���б� - �����б�
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com