��2013?�ֿ���һģ����������װ��ͼ���ش��й����⣺
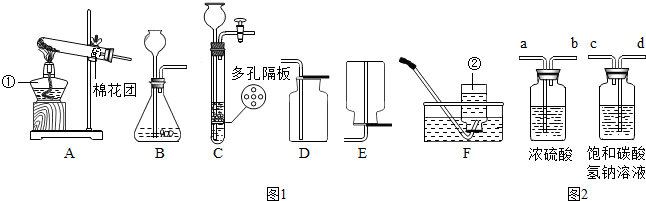
��1��ͼ1�б�����������ƣ���
�ƾ���
�ƾ���
����
����ƿ
����ƿ
��
��2������ͼ1��A��Dװ�õ���Ͽ�����ȡij�����壬ʵ������ȡ������Ļ�ѧ����ʽ��
2KMnO
4K
2M nO
4 +MnO
2 +O
2��
2KMnO
4K
2M nO
4 +MnO
2 +O
2��
������������
�ô����ǵ�ľ�����ڼ���ƿ�ڣ�����ȼ����
�ô����ǵ�ľ�����ڼ���ƿ�ڣ�����ȼ����
��
��3��ʵ����ͨ������ͼ1��װ��B��
D
D
�������ȡ������̼��ѡ����ĸ�����÷�Ӧ�Ļ�ѧ����ʽΪ
CaCO3+2HCl=CaCl2+CO2��+H2O
CaCO3+2HCl=CaCl2+CO2��+H2O
������Cװ�ô���Bװ�ý���ʵ�飬�ŵ�Ϊ
������ʱ���Ʒ�Ӧ�ķ�����ֹͣ
������ʱ���Ʒ�Ӧ�ķ�����ֹͣ
��
��Ϊ�˵õ�����������Ķ�����̼���壬����װ�ã���ͼ2���ĵ��ܰ�������������˳����
C
C
��ѡ����ĸ����
A��a��b��d��c B��b��a��c��d C��d��c��a��b D��c��d��b��a
�ڳ��ó���ʯ��ˮ���������̼�Ĵ��ڣ��÷�Ӧ�Ļ�ѧ����ʽΪ
CO2+Ca��OH��2=CaCO3��+H2O
CO2+Ca��OH��2=CaCO3��+H2O
��




 ������ϵ�д�
������ϵ�д� �żӾ���ϵ�д�
�żӾ���ϵ�д�
