
| 152-96-8 |
| 16 |
| 3.6g��2 |
| 18g/mol |
| 10.64L-1.68L |
| 22.4L/mol |
| 7.6g-0.4mol��1g/mol-0.4mol��12g/mol |
| 16g/mol |
 ���ڡ��䡢�Ծ��ɣ���
���ڡ��䡢�Ծ��ɣ��� �ȣ�
�ȣ�

 �Ͻ�ƽСѧ��������ϵ�д�
�Ͻ�ƽСѧ��������ϵ�д�
| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
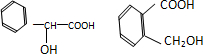
 ij�л���ķ��ӽṹ��ͼ��ͼ�й���ʾ������˫�������������������ĺ�����������ڸ��л����������ȷ���ǣ�������
ij�л���ķ��ӽṹ��ͼ��ͼ�й���ʾ������˫�������������������ĺ�����������ڸ��л����������ȷ���ǣ�������| A�����л���Ļ�ѧʽΪC6H6 | B�����л���ɷ���ȡ����Ӧ�ͼӳɷ�Ӧ | C�����л����ʹ��ˮ��ɫ����������NaOH��Һ��Ӧ | D�����л����������ͬϵ�� |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2012-2013ѧ�����ʡ���һ�и�һ6�½��Կ��Ի�ѧ�Ծ����������� ���ͣ������
����ij��A����Է�������Ϊ84���ش��������⣺
��1������������A�����������ϣ��������ʵ���һ�������ȼ������������������ȵ���(�����)________��
a��C7H12O2 b��C6H14 c��C6H14O d��C7H14O3
��2������AΪ���������������е�̼ԭ����ͬһƽ���ϣ��÷��ӵ�һ��ȡ����ֻ��һ�֡�A�Ľṹ��ʽΪ________________��
��3����A����ʹ��ˮ��ɫ������һ�ȴ���ֻ��һ�֣���A�Ľṹ��ʽΪ______________��
����1mol����ʽΪCmHn���л�������������ȫȼ�գ�����CO2��H2O�����ʵ�����ȣ�������168L����(�����)�����л�������ķ���ʽ____ _____��
����ij�л���A�����ĺ��������Ϊ�ⶨ�����������ʵ������
����һ���¶Ⱥ�ѹǿ�½�A��������������ͬ�¡�ͬѹ������������76����
�ڳ�ȡ7.6g�л���A���ڹ�����������ȫȼ�ա�����Ӧ��Ļ������ͨ������ŨH2SO4�������Ϊ10.64L��Ũ��������3.6g���ٽ����µ�����ͨ��ʢ��������ʯ�ҵĸ���ܣ��������������1.68L������������ڱ�״���²ⶨ���� ȷ��A�ķ���ʽ ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2015�����ʡ��һ6�½��Կ��Ի�ѧ�Ծ��������棩 ���ͣ������
����ij��A����Է�������Ϊ84���ش��������⣺
��1������������A�����������ϣ��������ʵ���һ�������ȼ������������������ȵ���(�����)________��
a��C7H12O2 b��C6H14 c��C6H14O d��C7H14O3
��2������AΪ���������������е�̼ԭ����ͬһƽ���ϣ��÷��ӵ�һ��ȡ����ֻ��һ�֡�A�Ľṹ��ʽΪ________________��
��3����A����ʹ��ˮ��ɫ������һ�ȴ���ֻ��һ�֣���A�Ľṹ��ʽΪ______________��
����1mol����ʽΪCmHn���л�������������ȫȼ�գ�����CO2��H2O�����ʵ�����ȣ�������168L����(�����)�����л�������ķ���ʽ____ _____��
����ij�л���A�����ĺ��������Ϊ�ⶨ�����������ʵ������
����һ���¶Ⱥ�ѹǿ�½�A��������������ͬ�¡�ͬѹ������������76����
�ڳ�ȡ7.6g�л���A���ڹ�����������ȫȼ�ա�����Ӧ��Ļ������ͨ������ŨH2SO4�������Ϊ10.64L��Ũ��������3.6g���ٽ����µ�����ͨ��ʢ��������ʯ�ҵĸ���ܣ��������������1.68L������������ڱ�״���²ⶨ���� ȷ��A�ķ���ʽ ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ���ӱ�ʡģ���� ���ͣ��ƶ���



�鿴�𰸺ͽ���>>
����ѧУ��ѡ - ��ϰ���б� - �����б�
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com