
�����Ϊ
2 L�������м���1 mol��N2��6 mol��H2���п��淴Ӧ��N2��3H2 2NH3��2 min����N2�����ʵ���Ϊ0.6 mol����
2NH3��2 min����N2�����ʵ���Ϊ0.6 mol����
(1)2 min�ڣ�N2�����ʵ���������0.4 mol��H2�����ʵ���������________mol��NH3��������������________mol��
(2)����N2��Ũ�ȱ仯����ʾ�÷�Ӧ�ķ�Ӧ����Ϊ________mol/(L��min)��
(3)����H2��Ũ�ȱ仯����ʾ�÷�Ӧ�ķ�Ӧ����Ϊ________mol/(L��min)��
(4)����NH3��Ũ�ȱ仯����ʾ�÷�Ӧ�ķ�Ӧ����Ϊ________mol/(L��min)��
(5)ͨ���������㣬����ʲô���֣�________________��

| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
��7�֣���һ�������£������Ϊ2 L�������м���2 mol O2��3 mol SO2ʹ֮��Ӧ����SO3���壺2SO2+O22SO3��2 minʱ�����O2�����ʵ���Ϊ1.6 mol����
��1��2min�ڣ�ƽ����Ӧ����v��O2���� �� v��SO3���� ��
��2��2minʱ��SO2��Ũ��Ϊ
��3����2minʱ���������Ӧ�ų�Q kJ����������д���÷�Ӧ���Ȼ�ѧ����ʽ
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
��9�֣���һ�������£������Ϊ2 L�������м���2 mol O2��3 mol SO2ʹ֮��Ӧ����SO3���壺2SO2+ O22SO3 ��2 minʱ�����O2�����ʵ���Ϊ1.6 mol����
��1��2min�ڣ�ƽ����Ӧ����v��O2���� �� v��SO3���� ��
��2��2minʱ��SO2��Ũ��Ϊ
��3����2minʱ���������Ӧ�ų�Q kJ����������д���÷�Ӧ���Ȼ�ѧ����ʽ
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2011-2012ѧ�긣��ʡ���嶫����ѧ��һ��ѧ�����п��Ի�ѧ�Ծ����������� ���ͣ������
��һ�������£������Ϊ2 L�������м���2 mol O2��3 mol SO2���п��淴Ӧ��
2SO2��g��+ O2��g�� 2SO3��g����2 min����O2�����ʵ���Ϊ1.6 mol����
2SO3��g����2 min����O2�����ʵ���Ϊ1.6 mol����
��1��2 min�ڣ�SO2�����ʵ��������� ��SO3�����ʵ��������� ��
��2������O2��Ũ�ȱ仯����ʾ�÷�Ӧ�ķ�Ӧ���ʣ���V��O2���� ��
��3������SO3��Ũ�ȱ仯����ʾ�÷�Ӧ�ķ�Ӧ���ʣ���V��SO3���� ����
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2012-2013ѧ���㽭ʡ��������������ѧ��һ5���¿��Ŀƻ�ѧ�Ծ����������� ���ͣ�������
��һ�������£������Ϊ2 L�������м���2 mol O2��3 mol SO2ʹ֮��Ӧ����SO3���壺2SO2+ O2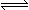 2SO3��2 minʱ�����O2�����ʵ���Ϊ1.6 mol����
2SO3��2 minʱ�����O2�����ʵ���Ϊ1.6 mol����
��1��2min�ڣ�ƽ����Ӧ����v��SO3����
��2��2minʱ��SO2��Ũ��Ϊ
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2015���㽭ʡ�����и�һ5���¿��Ŀƻ�ѧ�Ծ��������棩 ���ͣ�������
��һ�������£������Ϊ2 L�������м���2 mol O2��3 mol SO2ʹ֮��Ӧ����SO3���壺2SO2+
O2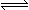 2SO3��2 minʱ�����O2�����ʵ���Ϊ1.6 mol����
2SO3��2 minʱ�����O2�����ʵ���Ϊ1.6 mol����
��1��2min�ڣ�ƽ����Ӧ����v��SO3����
��2��2minʱ��SO2��Ũ��Ϊ
�鿴�𰸺ͽ���>>
����ѧУ��ѡ - ��ϰ���б� - �����б�
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com