
������ʾ��þ�뱥��̼��������Һ��Ӧ������������Ͱ�ɫ�����ijͬѧ���������ʵ�鷽������֤���̽����Ӧԭ����
(1)�������
ʵ�����ɰֽ��ȥþ����������Ĥ���������ʢ���������з�̪�ı���̼��������Һ���Թ��У�Ѹ�ٷ�Ӧ�������������ݺͰ�ɫ�������Һ��dz���졣
��ͬѧ�Է�Ӧ�в����İ�ɫ�������������²²⣺
�²�1����ɫ���������Ϊ________________��
�²�2����ɫ���������ΪMgCO3��
�²�3����ɫ����������Ǽ�ʽ̼��þ[xMgCO3��yMg(OH)2]��
(2)��ƶ���ʵ��ȷ�����ﲢ��֤�²⣺
| ʵ����� | ʵ�� | ʵ������ | ���� |
| ʵ��� | ��ʵ������ռ����������ȼ | �ܰ���ȼ�ա���������ɫ���� | ������ɷ�Ϊ________ |
| ʵ��� | ��ȡʵ����еİ�ɫ�����ϴ�ӣ���������________ | ��________________ __________________ __________________ | ��ɫ��������ܺ���MgCO3 |
| ʵ��� | ȡʵ����еij���Һ�������м�������CaCl2ϡ��Һ | ������ɫ���� | ����Һ�д���________ |

��(1)Mg(OH)2
(2)����������ϡ����(��������)���۲������ݣ�����ȫ���ܽ⡡��CO32��
(3)2MgCO3��Mg(OH)2��Mg(OH)2��2MgCO3��Mg3(OH)2(CO3)2
(4)NaHCO3��Һ�д���ƽ�⣺HCO32�� H����CO32����H2O
H����CO32����H2O H����OH����Mg��H����Ӧ����H2��Mg2����Mg2����OH����CO32����Ӧ����������Mg(OH)2��2MgCO3����H����OH����CO32����Ũ�Ⱦ���С����ʹ������ƽ��������ƶ�
H����OH����Mg��H����Ӧ����H2��Mg2����Mg2����OH����CO32����Ӧ����������Mg(OH)2��2MgCO3����H����OH����CO32����Ũ�Ⱦ���С����ʹ������ƽ��������ƶ�
����



| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ʵ����
��������һ����Ҫ���л�����ԭ�ϣ��Ʊ��������ԭ����95%�Ҵ���80%���ᣨ������ˮϡ��Ũ���ᣩ����ϸ���廯�Ʒ�ĩ�ͼ������Ƭ���÷�Ӧ��ԭ�����£�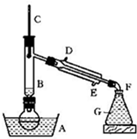
NaBr + H2SO4 �� NaHSO4 + HBr
CH3CH2OH + HBr CH3CH2Br + H2O
CH3CH2Br + H2O
ij����С������ʵ�����Ʊ��������װ������ͼ���������±���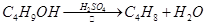
| ���� ���� | �Ҵ� | ������ | 1��2-�������� | ���� | Ũ���� |
| �ܶ�/g��cm-3 | 0.79 | 1.46 | 2.2 | 0.71 | 1.84 |
| �۵㣨�棩 | ��130 | ��119 | 9 | ��116 | 10 |
| �е㣨�棩 | 78.5 | 38.4 | 132 | 34.6 | 338 |
| ��ˮ�е��ܽ�ȣ�g/100gˮ�� | ���� | 0.914 | 1 | 7.5 | ���� |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ʵ����
���������[(NH4)2SO4��FeSO4��6H2O]Ϊdz��ɫ���壬������ˮ�������ھƾ�����ˮ�е��ܽ�ȱ�FeSO4��(NH4)2SO4��ҪС��ʵ�����г��Է���мΪԭ�����Ʊ����䲽�����£�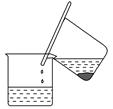
ͼ1
����1����м�Ĵ�����������м�����ȵ�̼������Һ�н��ݼ����Ӻ���ͼ1��ʾ������������岢ϴ�ӡ����
����2��FeSO4��Һ���Ʊ����������õ���м������ƿ�����������3 mol��
L��1H2SO4��Һ����������ַ�ӦΪֹ�����ȹ���(��ͼ2��ʾ)���ռ���Һ��ϴ��Һ��
ͼ2
����3����������淋��Ʊ���������FeSO4��Һ�м��뱥��(NH4)2SO4��Һ����������Ũ������ȴ�ᾧ�����ˡ��Ҵ�ϴ�Ӻ�õ���������茶��塣
��ش��������⣺
(1)����1��ͼ1���뷽����Ϊ________����
(2)����2����һ�����Բ���������___________________________________��
���ȹ��˵�������________________________________________________��
(3)����3����Ũ�������У���________ʱֹͣ���ȡ�����ˮ�Ҵ�ϴ�Ӿ����ԭ����______________________________________________________________��
(4)FeSO4��7H2O�ڳ�ʪ�Ŀ������ױ�������Fe(OH)SO4��3H2O��д���÷�Ӧ�Ļ�ѧ����ʽ________________________________________________________��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ʵ����
��˾ƥ�ֿ���ˮ�����������������Ƶá����Ʊ�ԭ�����£�

��˾ƥ��(����ˮ����)������������ˮ����˾ƥ�ֿɰ����²�����ȡ�ʹ�����
����1���ڸ����50 mLԲ����ƿ�м���2 gˮ���ᡢ5 mL��������5��Ũ���ᣬ��ʹˮ����ȫ���ܽ⡣
����2����ͼ��ʾװ��װ���������ͨˮ����ˮԡ�ϼ��Ȼ���5��10 min������ˮԡ�¶���85��90 �档
����3����Ӧ������ȡ�·�Ӧƿ����ȴ���ٷ����ˮ����ȴ���ᾧ�����ˡ���ˮϴ��2��3�Σ��������˵ôֲ��
����4�����ֲ���ת����150 mL�ձ��У��ڽ����¼���25 mL����̼��������Һ����ֽ��裬Ȼ����ˡ�
����5������Һ����10 mL 4 mol��L��1���ᣬ���裬���ձ����ڱ�ԡ����ȴ��ʹ�ᾧ��ȫ�����ˣ�������ˮϴ��2��3�Ρ�
(1)����1Ũ��������ÿ�����________��
(2)����2�У�������ͨˮ��ˮӦ��________�ڽ�(�a����b��)��
(3)����3����ʱ����ʱ��ֽ�ᴩ�ף�������ֽ���Ĵ�ʩ��______________________________________________________________��
(4)����4������Ҫ��Ӧ�Ļ�ѧ����ʽΪ_____________________�����˵õ��Ĺ���Ϊ________��
(5)ȡ��������5��õľ������ʢ��5 mLˮ���Թ��У�����1��2��1%���Ȼ�����Һ��������Һ����ɫ���ɲ���________��������һ���������塣
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ʵ����
SiO2��SO2��CO2����������������ǵĻ�ѧ���ʾ���һ���������ԣ�Mg��Na�Ļ�ѧ����Ҳ����һ�������ԡ�
ij��ȤС������ͼ��ʾװ�ý���Mg��SO2��Ӧ��ʵ�顣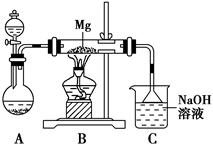
(1)ѡ����ȡSO2�ĺ����Լ�________(����)��
��ŨHCl����ŨH2SO4����Na2SO3���塡��CaSO3����
(2)����װ�û����Ż����Ż��ķ�����________________________________________��װ��C��NaOH��Һ��������___________________________________________________________
(3)��ͬѧ�Ʋ�Mg��SO2�ķ�Ӧ��Mg��CO2�ķ�Ӧ���ƣ���÷�Ӧ����ʽΪ_________________________________________��
��ͬѧ���Ʋ��ǣ�2Mg��3SO2 2MgSO3��S����ͬѧ���Ʋ��ǣ�3Mg��SO2
2MgSO3��S����ͬѧ���Ʋ��ǣ�3Mg��SO2 2MgO��MgS��Ҫ��֤�ס��ҡ�����λͬѧ���Ʋ��Ƿ���ȷ����ͬѧ������ʵ��̽����
2MgO��MgS��Ҫ��֤�ס��ҡ�����λͬѧ���Ʋ��Ƿ���ȷ����ͬѧ������ʵ��̽����
��֪��MgSO3��MgS������ˮ���������ᷢ�����ֽⷴӦ�ų����壻H2S����ͨ��CuSO4��Һ�г��ֺ�ɫ������
��ѡ�Լ���2 mol��L��1���ᡢ2 mol��L��1���ᡢ����ˮ��2 mol��L��1 NaOH��Һ��Ʒ����Һ������ʯ��ˮ��2 mol��L��1 CuSO4��Һ����������Ʒ��ѡ��
| ��� | ʵ�鲽�� | Ԥ������ͽ��� |
| �� | ȡ������Ӧ�����ù������Թ��� | |
| �� | ���Թ��еĹ��������μ�____________���Թܿ����ϴ����ܵĵ���������������ͨ��ʢ��________���Թ��� | ���Թ��е�________�����ͬѧ�Ʋ���ȷ�����Թ��еĹ���δ��ȫ�ܽ⣬��________������ͬѧ�Ʋ���ȷ |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ʵ����
�������Ʊ���Ϊ��ҵ�Σ���Ư�ס���Ƶȷ���Ӧ�ù㷺����ľ̿��Ũ���ᡢˮ��ͭΪԭ�����ɵ�һ��������������Ʒ�Ӧ�Ʊ��������Ƶ�װ������ͼ��ʾ�����ּг�װ���ԣ���
��֪�������£���2NO+Na2O2��2NaNO2 ��3NaNO2+3HCl��3NaCl+HNO3+2NO��+H2O��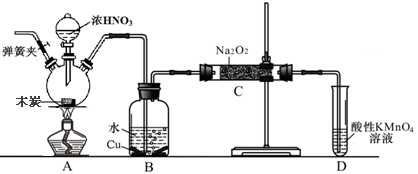
�����������£�NO��NO2�C������MnO4�C��Ӧ����NO3�C��Mn2+
���������գ�
��1��д��Ũ������ľ̿��Ӧ�Ļ�ѧ����ʽ ��
��2��B�й۲쵽����Ҫ������ ��Dװ�õ������� ��
��3������C�в������������Ƶķ����� ��
��4��������C�����г�������������и�����̼���ƺ� ��Ϊ���������Щ������Ӧ��B��Cװ�ü�����װ��E������E��ע��E��ʢ�ŵ�ҩƷ���� ��
��5��д������C�������Ƿ�̼���Ƶķ��� ��
��6����1.56g����������ȫת����Ϊ�������ƣ�������������Ҫľ̿ g��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ʵ����
�������ƣ�NaClO2����һ�ָ�Ч��������Ư������֪��NaClO2������Һ���¶ȵ���38��ʱ�����ľ�����NaClO2��3H2O������38��ʱ����������NaClO2������60��ʱNaClO2�ֽ��NaClO3��NaCl��������ͼ��ʾװ���Ʊ��������ơ�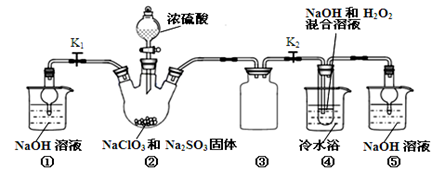
���������գ�
��1��װ�â��в���ClO2�Ļ�ѧ����ʽΪ ��װ�â۵������� ��
��2����װ�âܷ�Ӧ�����Һ���NaClO2����IJ�������Ϊ��
�ټ�ѹ��55�������ᾧ���� ���� ���ܵ���60�����õ���Ʒ��
��3��ȷ��ȡ��������������Ʒ10g���ձ��У�������������ˮ�����ĵ⻯�ؾ��壬�ٵ���������ϡ���ᣬ��ַ�Ӧ��ClO2��+ 4I��+4H+ ��2H2O+2I2+Cl�����������û��Һ���250mL������Һ�����ƴ���Һ���õ��Ķ������������� ��
��4��ȡ25.00mL����Һ����2.0 mol/L Na2S2O3��Һ�ζ���I2 +2S2O32����2I��+S4O62�������Ե�����Һ��ָʾ�����ﵽ�ζ��յ�ʱ������Ϊ ���ظ��ζ�2�Σ����Na2S2O3��Һƽ��ֵΪ20.00 mL������Ʒ��NaClO2����������Ϊ ��
��5��ͨ������˵��װ�â��ڱ�ʵ���е����� ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ʵ����
�о���ѧϰС���ij�����������壨FeSO4?xH2O���ȷֽ��о�����С��ͬѧ��ȡag��������������Ʒ��ͼ1���¼��ȣ�ʹ����ȫ�ֽ⣬�����ò������̽������ͨ������װ��B�������x��ֵ��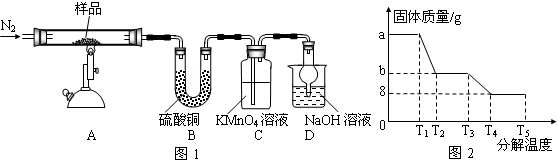
��1��װ��B������ͭ��ĩ��������������12.6g��˵����������ˮ��װ��C�и��������Һ��ɫ��˵�������л��� ��
��2��ʵ����Ҫ����ͨ�뵪������������x�� ���ƫ����ƫС�����䡱����
��3����������������ȫ�ֽ��װ��A�л���������ɫ����Fe2O3��
��4���������Ϸ����ó����������ֽ������һ����SO3��д��FeSO4�ֽ�Ļ�ѧ����ʽ ��
��5��װ��D���θ���ܵ����� ��
��6��ij�о�������SDTQ600�ȷ����Ƕ������������壨FeSO4?xH2O�������ȷֽ⣬���������ݣ����Ƴɹ������������ֽ��¶ȵĹ�ϵͼ��ͼ2������ͼ2���й����ݣ��ɼ����FeSO4?xH2O�е�x= ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ʵ����
���������纣���������к��зḻ�ĵ�Ԫ�أ���Ҫ�Ե⻯����ʽ���ڡ���һ��ѧ����С���ú���Ϊԭ����ȡ�����ⵥ�ʣ����ǽ��������ճɻң���ˮ����һ��ʱ��(���õ⻯�����ܽ���ˮ��)���õ�����������Һ��Ȼ������ʵ��������ȡ���ʵ⣺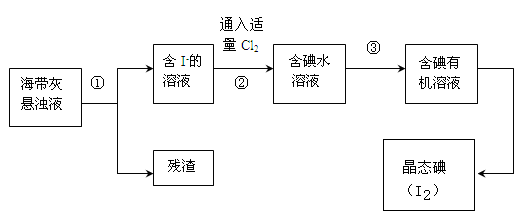
��1��ָ����ȡ��Ĺ������йص�ʵ��������ƣ���_________����________��
��2�����̢��г�������Cl2��Ӧ�����ӷ���ʽΪ_____________________��
��3�������������õ��л��Լ�������___________(ֻ��һ��)������ѡ��������__________________��
��4���Ӻ�����л���Һ����ȡ��ͻ����л���Һ������Ҫ�������۲���ͼ��ʾʵ��װ�ã�ָ�������֮�������Ը���_______________________________________________________��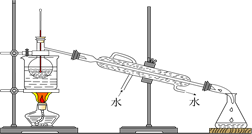
(5)���������������ʱ��ʹ��ˮԡ��ԭ����______________________��
�鿴�𰸺ͽ���>>
����ѧУ��ѡ - ��ϰ���б� - �����б�
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com