��2010?Ϋ��һģ���о���ѧ��Ӧԭ���������������Ǻ�������ģ�
��1�����й��ڴ����������ȷ����
abd
abd
����д���ţ���
a���������������ᣬ��ʳ����Ҫ�ɷ�
b���������Һ�м���һ����NaOH���壬��Һ�ĵ�������ǿ
c����ˮϡ�ʹ�����Һ����Һ�е���������Ũ�Ⱦ���С
d�������£�������Һ��ˮ�ĵ���̶ȼ�С
��2�������ʶ��������ű������ã�����Ҫ�ɷ�ΪCa
5��PO
4��
3OH����������ˮ�д��ڳ����ܽ�ƽ�⣬��д�������ʵ��ܶȻ���������ʽK
ap=
c��Ca2+��5c��PO43-��3c��OH-��
c��Ca2+��5c��PO43-��3c��OH-��
��
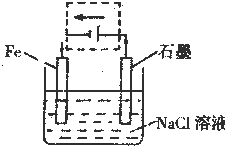
��3���������������洦�ɼ��������ĵ绯��ʴԭ����ͼ��ʾ��
��д��ʯī�缫�ĵ缫��Ӧʽ
O2+4e-+2H2O=4OH-
O2+4e-+2H2O=4OH-
��
�ڽ���װ�������ļ��ɳ�Ϊ�����绯ѧ������װ�ã�������ͼ���߿�����ʾλ�������ģ����ü�ͷ��������е�����������
��д���ĺ�ʯī�缫�ĵ缫��Ӧʽ
2Cl-2e-=Cl2��
2Cl-2e-=Cl2��
��

 ��2010?Ϋ��һģ���о���ѧ��Ӧԭ���������������Ǻ�������ģ�
��2010?Ϋ��һģ���о���ѧ��Ӧԭ���������������Ǻ�������ģ�