

| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ��2013��������·ʵ����ѧ�߶���ѧ����ĩ���Ի�ѧ�Ծ� ���ͣ�ѡ����
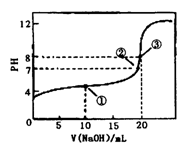
�����£���0.100 0 mol��L��1 NaOH��Һ�ζ�20.00 mL 0.100 00 mol��L��1 CH3COOH��Һ�ζ�������ͼ��ʾ������˵����ȷ����

A�������ʾ��Һ�У�c(CH3COO��)��c(OH��)��c(CH3COOH)��c(H��)
B�������ʾ��Һ�У�c(Na��)��c(CH3COOH)��c(CH3COO��)
C�������ʾ��Һ�У�c(Na��)>c(OH��)>c(CH3COO��)>c(H��)
D���ζ������п��ܳ��֣�c(CH3COOH)>c(CH3COO��)>c(H��)>c(Na��)>c(OH��)
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ���㽭ʡģ���� ���ͣ�ʵ����

�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�����ո߿����� ���ͣ�������ѡ����

�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��������ĩ�� ���ͣ���ѡ��

�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
�����£���0.100 0 mol��L��1 NaOH��Һ�ζ�20.00 mL 0.100 00 mol��L��1 CH3COOH��Һ�ζ�������ͼ��ʾ������˵����ȷ����
A�������ʾ��Һ�У�
c(CH3COO��)��c(OH��)��c(CH3COOH)��c(H��)
B�������ʾ��Һ�У�c(Na��)��c(CH3COOH)��c(CH3COO��)
C�������ʾ��Һ�У�c(Na��)>c(OH��)>c(CH3COO��)>c(H��)
D���ζ������п��ܳ��֣�
c(CH3COOH)>c(CH3COO��)>c(H��)>c(Na��)>c(OH��)

�鿴�𰸺ͽ���>>
����ѧУ��ѡ - ��ϰ���б� - �����б�
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com