


 ϰ�⾫ѡϵ�д�
ϰ�⾫ѡϵ�д�
| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
| ������� | �ռ����� | ������� | ��֤���� | �ó����� |
| ��ˮ�к���������ʹʪ��ĺ�ɫ������ɫ | ��������ǿ������ ����������ˮ��Ӧ��������ʹ����� �۴�������ǿ������ |
�� �� �� ��H2Oʹ������ɫ |
ʵ��٣��Ѻ�ɫ�ɲ���������������ļ���ƿ�У���������ɫ ʵ��ڣ� ʵ��ۣ� ʵ��ܣ��Ѻ�ɫ��������ˮ�ﲼ������ɫ |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ��Ķ�����
������ˮ�к��ж������ӣ�ijУ��ѧ�о���ѧϰС���ͬѧΪ̽�������ʣ���������ʵ��,���������ɣ�
��1��������ʹʪ��ĺ�ɫ������ɫ��ʹ����ɫ�����Ļ�ѧʽ��______
��2������ˮ�ڹ�����һ��ʱ�䣬��Һ��ɫ��dz�����йط�Ӧ�Ļ�ѧ����ʽΪ��
��
��3��ƽ�ⳣ����K�������˷����ϵ�Ŀ��淴Ӧ�ڸ������¶��½��еij̶ȣ�����ͬһ�����͵ķ�Ӧ��ƽ�ⳣ��Խ������Ӧ���еij̶�Խ��
H2CO3![]() HCO3-�� H�� Ka1(H2CO3)=4.45��10-7
HCO3-�� H�� Ka1(H2CO3)=4.45��10-7
HCO3-![]() CO32-��H�� Ka2(HCO3-)=5.61��10-11
CO32-��H�� Ka2(HCO3-)=5.61��10-11
HClO![]() H����ClO- Ka(HClO)=2.95��10-8
H����ClO- Ka(HClO)=2.95��10-8
���������ϵ���ƽ�ⳣ������д��������������ͨ�뵽������̼������Һ����������Ӧ�����ӷ���ʽ��
��4��������ˮ��ʯ��ʯ�ķ�Ӧ����ȡ��ŨHClO��Һ�ķ���֮һ��
ʵ��һ�������о���
�����Թ��м�������Ŀ�״̼��ƣ��ټ���Լ20mL������ˮ����ַ�Ӧ�� ���������ݲ�������Һdz����ɫ��ȥ��
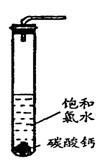 �ڹ��ˣ�����Һ������ɫ�����ϣ����������ˮ��Ư���Ը�ǿ��
�ڹ��ˣ�����Һ������ɫ�����ϣ����������ˮ��Ư���Ը�ǿ��
��Ϊ��ȷ����Ӧ�������Һ��Ϊ���ݣ��ֱ��������ʵ�飺
��һ����ʯ��ˮ��ϣ���������������ɫ������
�ڶ�����ϡ�����ϣ���������������ɫ���壻
�������ݼ��ȣ�������Һ��������д�����ɫ���������
����⣬����ʵ���в�������ɫ�����ΪCO2��
��ش�
�ٷ�Ӧ�����õ���ҺƯ������ǿ��ԭ����______ ___________ ____
����������ʵ�����֪���ڵ���Һ�е����ʳ�CaCl2��HClO�⣬������______ _ ��
ʵ����������о���
��Բ����ƿ�ײ�����һ����������ס�Ĺ�����״ ̼��ƺ�150mL������ˮ������ͼ��ʾװ��ʵ�飬�����ٲ������ݺ���������ʣ���ʯ��ʯ���Һ�棬�ܷ���ټ��ȡ������ƿ�е�Һ�壬ֱ����Ͳ�е����岻�����ӣ�������HClO�ķֽ⣩��
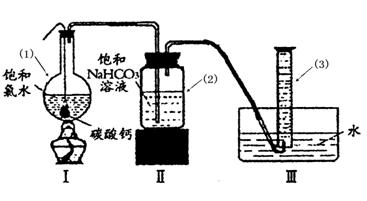
��ش�
��д��������1����������3�������ƣ���1�� ��3��
��Ϊ������װ�â��ռ����������CO2���ܽ����ɵ���ʧ����ˮ������ȻΪˮ�������װ�â���иĽ�����ķ����� ��
�����ȷ������Ͳ����������
a��________ _ ________ _ ________ _ ________
b�������ƶ���Ͳ����Ͳ��Һ����ˮ��Һ����ƽ
c�� ________ _ ________
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ��Ķ�����
������ˮ�к��ж�������,ijУ��ѧ�о���ѧϰС���ͬѧΪ̽��������,��������ʵ��,���������ɣ�
��1��������ʹʪ��ĺ�ɫ������ɫ��ʹ����ɫ�����Ļ�ѧʽ��______
��2������ˮ�ڹ�����һ��ʱ�䣬��Һ��ɫ��dz�����йط�Ӧ�Ļ�ѧ����ʽΪ��
��
��3��ƽ�ⳣ�������˷����ϵ�Ŀ��淴Ӧ�ڸ������¶��½��еij̶ȣ�����ͬһ�����͵ķ�Ӧ��ƽ�ⳣ��Խ������Ӧ���еij̶�Խ��
H2CO3
��H�� Ka1��H2CO3��=4.45��10��7
��H�� Ka2(HCO3��)=5.61��10��11
HclO H����
Ka(HClO)=2.95��10��8
���������ϵ���ƽ�ⳣ������д��������������ͨ�뵽������̼������Һ����������Ӧ�����ӷ���ʽ����
��4��������ˮ��ʯ��ʯ�ķ�Ӧ����ȡ��ŨHClO��Һ�ķ���֮һ��
ʵ��һ�������о���
�� ���Թ��м�������Ŀ�״̼��ƣ��ټ���Լ20mL������ˮ����ַ�Ӧ��
���������ݲ�������Һdz����ɫ��ȥ��
�� ���ˣ�����Һ������ɫ�����ϣ����������ˮ��Ư���Ը�ǿ��
�� Ϊ��ȷ����Ӧ�������Һ��Ϊ���ݣ��ֱ��������ʵ�飺
��һ����ʯ��ˮ��ϣ���������������ɫ������
�ڶ�����ϡ�����ϣ���������������ɫ���壻
�������ݼ��ȣ�������Һ��������д�����ɫ���������
����⣬����ʵ���в�������ɫ�����ΪCO2��
��ش�
�� ��Ӧ�����õ���ҺƯ������ǿ��ԭ����______ ___________ ____
����������ʵ�����֪���ڵ���Һ�е����ʳ�CaCl2��HClO�⣬������______ _ ��
ʵ����������о���
��Բ����ƿ�ײ�����һ����������ס�Ĺ�����״ ̼��ƺ�150mL������ˮ������ͼ��ʾװ��ʵ�飬�����ٲ������ݺ���������ʣ���ʯ��ʯ���Һ�棬�ܷ���ټ��ȡ������ƿ�е�Һ�壬ֱ����Ͳ�е����岻�����ӣ�������HClO�ķֽ⣩��
��ش�
�� �������1����������3��������
��1�� ��3��
�� Ϊ������װ�â��ռ����������CO2���ܽ����ɵ���ʧ����ˮ������ȻΪˮ�������װ�â���иĽ�����ķ����� ��
�� ���ȷ������Ͳ����������
a________ _
b �����ƶ���Ͳ����Ͳ��Һ����ˮ��Һ����ƽ
c
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2011�콭��ʡ�Ž��и���2����У���������ۣ���ѧ���� ���ͣ�ʵ����
������ˮ�к��ж�������,ijУ��ѧ�о���ѧϰС���ͬѧΪ̽��������,��������ʵ��,���������ɣ�
��1��������ʹʪ��ĺ�ɫ������ɫ��ʹ����ɫ�����Ļ�ѧʽ��______
��2������ˮ�ڹ�����һ��ʱ�䣬��Һ��ɫ��dz�����йط�Ӧ�Ļ�ѧ����ʽΪ��
��
��3��ƽ�ⳣ�������˷����ϵ�Ŀ��淴Ӧ�ڸ������¶��½��еij̶ȣ�����ͬһ�����͵ķ�Ӧ��ƽ�ⳣ��Խ������Ӧ���еij̶� Խ��
Խ��
H2CO3 
 �� H�� Ka1��H2CO3��=4.45��10��7
�� H�� Ka1��H2CO3��=4.45��10��7

 ��H�� Ka2(HCO3��)=5.61��10��11
��H�� Ka2(HCO3��)=5.61��10��11
HclO  H����
H���� Ka(HClO)=2.95��10��8
Ka(HClO)=2.95��10��8
���������ϵ���ƽ�ⳣ������д��������������ͨ�뵽������̼������Һ����������Ӧ�����ӷ���ʽ����
��4��������ˮ��ʯ��ʯ�ķ�Ӧ����ȡ��ŨHClO��Һ�ķ���֮һ��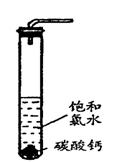
ʵ��һ�������о���
�� ���Թ��м�������Ŀ�״̼��ƣ��ټ���Լ20mL������ˮ����ַ�Ӧ��
���������ݲ�������Һdz����ɫ��ȥ��
�� ���ˣ�����Һ������ɫ�����ϣ����������ˮ��Ư���Ը�ǿ��
�� Ϊ��ȷ����Ӧ�������Һ��Ϊ���ݣ��ֱ��������ʵ�飺
��һ����ʯ��ˮ��ϣ���������������ɫ������
�ڶ�����ϡ�����ϣ���������������ɫ���壻
�������ݼ��ȣ�������Һ��������д�����ɫ���������
����⣬����ʵ���в�������ɫ�����ΪCO2��
��ش�
�� ��Ӧ�����õ���ҺƯ������ǿ��ԭ����______ ___________ ____
����������ʵ�����֪���ڵ���Һ�е����ʳ�CaCl2��HClO�⣬������_______ ��
ʵ����������о���
��Բ����ƿ�ײ�����һ����������ס�Ĺ�����״ ̼��ƺ�150mL������ˮ������ͼ��ʾװ��ʵ�飬�����ٲ������ݺ���������ʣ���ʯ��ʯ���Һ �棬�ܷ���ټ��ȡ������ƿ�е�Һ�壬ֱ����Ͳ�е����岻�����ӣ�������HClO�ķֽ⣩��
�棬�ܷ���ټ��ȡ������ƿ�е�Һ�壬ֱ����Ͳ�е����岻�����ӣ�������HClO�ķֽ⣩��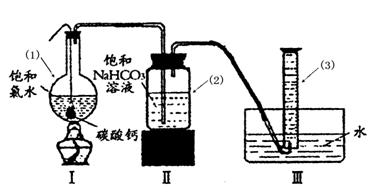
��ش�
���������1����������3��������
��1�� ��3��
��Ϊ������װ�â��ռ����������CO2���ܽ����ɵ���ʧ����ˮ������ȻΪˮ�������װ�â���иĽ�����ķ����� ��
�����ȷ������Ͳ����������
a________ _
b �����ƶ���Ͳ����Ͳ��Һ����ˮ��Һ����ƽ
c
�鿴�𰸺ͽ���>>
����ѧУ��ѡ - ��ϰ���б� - �����б�
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com