
ʳ�����ճ�����ı���Ʒ��Ҳ����Ҫ�Ļ���ԭ�ϡ�
��1����ʳ�γ���������K+��Ca 2+��Mg 2+��Fe 3 + ��![]() ���������ӣ�ʵ�����ᴿNaCl���������£�
���������ӣ�ʵ�����ᴿNaCl���������£�

�ṩ���Լ�������Na2CO3��Һ ����K2CO3��Һ NaOH��Һ BaCl2 ��Һ
Ba��NO3��2��Һ 75���Ҵ� ���Ȼ�̼
������ȥ��Һ���е�Ca 2+��Mg 2+��Fe 3 + ��![]() ���ӣ�ѡ��a���������Լ������μ�˳������Ϊ
���ӣ�ѡ��a���������Լ������μ�˳������Ϊ
��ֻ�ѧʽ����
��ϴ�ӳ�ȥNaCl������渽��������KCl��ѡ�õ��Լ�Ϊ ��
��2�����ᴿ��NaCl����500 mL4��00 mol? L-1NaCl��Һ������������ҩ�ס����������
�����������ƣ���
 �Ķ��쳵ϵ�д�
�Ķ��쳵ϵ�д�
| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
��1��ը����ʱ1 kg��������0.5 kgˮ��4 g������10 gС�մ�����ʳ�εȸ��ϣ�����ը����Ʒ�����IJ���һ��Ϊ80%��ͨ������˵����ÿ��ʳ��100 g��������������������__________________��?
��2�����о��ҹ��������ճ�����������������ʳƷ���Ӽ��⣩�����ֿ���;����______________________________________________________________________?
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��0115 �¿��� ���ͣ������
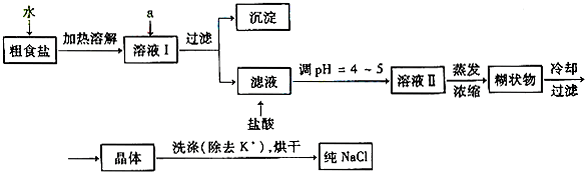
�鿴�𰸺ͽ���>>
����ѧУ��ѡ - ��ϰ���б� - �����б�
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com