
��ѧ��ȤС���ͬѧΪ�ⶨijNa2CO3��NaCl�Ĺ���������Ʒ��Na2CO3��������������������ʵ�飬������벢��ɶ��й�����Ľ��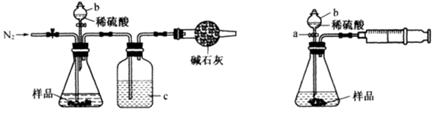
ͼ1 ͼ2
��1����ͬѧ��ͼ1��ʾװ�òⶨCO2��������ʵ��ʱϡ����������Ʒ�е� (�Na2CO3����"NaCl��)������Ӧ������b�������� ��ϴ��ƿc��ʢװ����Ũ���ᣬ��Ũ����������� ��
��2����ͬѧ��ͼ2��ʾװ�ã�ȡһ����������Ʒ(Ϊm g���Ѳ��)������ϡ���ᷴӦ����ʵ�飬�����Ʒ��Na2CO3�����������IJⶨ��
��ʵ��ǰ������װ�������Եķ������ȴ���a����bע��ˮ�����¶˲��������γ�һ��ˮ�����ٽ���Ͳ����������ѹ����b�¶˲������е� ��������װ�����������á�
����ʵ�����ʱ����ֱ�Ӳ�õ�������CO2�� (������������������)��
��3����ͬѧ����ͼ��ʾ�����Ͳ���ʵ�飺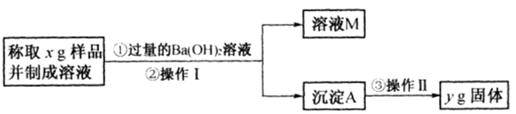
�ٲ���I�漰��ʵ�������� ��ϴ�ӣ��������漰��ʵ�������и�� ��
�ڱ���õ���Ʒ��Na2CO3���������ļ���ʽΪ ��
��4����״���£���672 mL CO2��ͨ��50 mL1mol/LKOH��Һ�У���ȫ��Ӧ��������Һ��K2CO3��KHCO3�����ʵ���֮��Ϊ(�跴Ӧǰ����Һ����仯���Բ���) ��
��1��Na2CO3 (1��) ��Һ©��(1��) ��ȥCO2�е�ˮ����(1��)
��2����Һ��(1��)�����(1��)
��3���ٹ���(1��) ����(1��) ��106y/197x (1��)
��4��n(K2CO3):n(KHCO3)=2:1(2��)
���������������1�� NaCl����ϡ���ᷴӦ����ѡNa2CO3������b�������Ƿ�Һ©����Ũ����������dz�ȥCO2�е�ˮ�����������CO2���壩����2�� �ٽ���Ͳ����������ѹ�������������е�ѹǿ������b�¶˲������е�Һ����������װ�����������á���CO2�����壬����ֱ�Ӳ�õ�������CO2���������3�� �ٳ������ɣ��ʲ���I��Ҫ�漰���˲�����Ҫ֪�������������Ҫ���ء��ھ�����ѧʽ�ļ��㣬��Ʒ��Na2CO3���������ļ���ʽΪ106y/197x����4�������ķ�ӦΪ0.03CO2+0.05KOH=XK2CO3+YKHCO3+0.025H2O��X+Y=0.03��2X+Y=0.05�����X=0.02��Y=0.01������Һ��K2CO3��KHCO3�����ʵ���֮��Ϊn(K2CO3):n(KHCO3)0.02��0.01=2:1��
���㣺���⿼���ȥijNaCl��Ʒ�е�����Na2CO3����Ʒ��Na2CO3�������������漰����ԭ������ѧ̽�����̣���һ���ۺ���ǿ�ĺ��⡣



| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ʵ����
������������(FeC2O4��2H2O)���������Լ�����Ӱ�������͵�ز�����������﮵��������ش��������⣺
I����ȤС��Բ�����������ķֽ�������ʵ���̽����̽���ֽ�õ��Ĺ����������Ԫ�صĴ�����ʽ��
��1���������
����һ��___________�� �������ȫ����FeO �� ��������FeO��Fe����
��2�����ʵ�鷽��֤����������
| ʵ�鲽�� | ��������� |
| ����1�����Թ��м��������������ټ������� ������� | ����Һ��ɫ���Ըı࣬���� ���ɣ���֤���������ʴ��� |
| ����2��������1�еõ�����Һ���ˣ���������ˮϴ����ϴ��Һ��ɫ | |
| ����3��ȥ����2�õ������������Թ��У��μ� | |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ʵ����
��������(Na2O2)����ѧ�������ʡ���֪������������CO2��Ӧ���������ɣ�����SO2ͨ��������Ʒ�ĩ��Ҳ���������ɡ��������CO2��SO2��������Ƶķ�Ӧԭ����ͬ����Ҳ�������SO2���н�ǿ�Ļ�ԭ�ԣ��ܱ����������������������ƣ�CO2��ǿ��ԭ�ԣ���Ӧԭ������ͬ���ݴ��������ʵ����������жϡ�
ʵ��һ����һ�����Ĺ������ƹ�����ͨ��������SO2��ȡ��Ӧ��Ĺ������ʵ��̽������֤������������SO2��Ӧ���ص㡣
��1��������裺����1����Ӧ�������ֻ��________��֤��SO2δ��������
����2����Ӧ�������ֻ��________��֤��SO2��ȫ��������
����3��__________________________֤��______________________��
ʵ��̽����ʵ�����ͨ���������������жϷ����Ļ�ѧ��Ӧ��ʵ��װ�����£�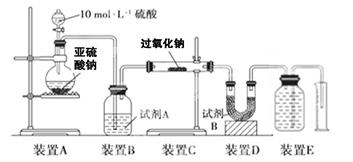
��2���Լ�A����ѡ��________���Լ�B��������________��
��3��ʵ����װ��C�й�����������������m1 g��װ��D����������m2 g��װ��E���ռ���������ΪV L(�ѻ���ɱ�״����)���������йز��������жϣ� SO2δ������ʱ����ȫ��������V-m1��ϵʽ��
δ��������____________����ȫ��������____________��
��4����SO2��ȫ��������д����Ӧ�Ļ�ѧ����ʽ�� __________________________��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ʵ����
ij̽��С�������ͼ��ʾװ�ý���Fe����ˮ�����ķ�Ӧ��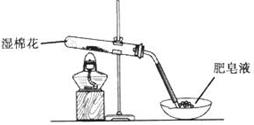
��1��ʵ��ǰ���װ�������Եķ���Ϊ________________________________________________________��
��2������ʵ�������������ʵ�������_____________________________________________��
��3����̽��С���Ϊ���飬����ͼװ�ý��жԱ�ʵ�飬�����þƾ���ơ������þƾ��Ƽ��ȣ���Ӧ�����Ϊ��ɫ��ĩ(������)������ֱ��ò����������ʵ�顣
| ���� | ���� | �������� | �������� |
| 1 | ȡ��ɫ��ĩ����ϡ���� | �ܽ⣬������ | �ܽ⣬������ |
| 2 | ȡ����1����Һ���μ�����KMnO4��Һ | ��ɫ��ȥ | ��ɫ��ȥ |
| 3 | ȡ����1����Һ���μ�KSCN��Һ | ��� | ������ |
| 4 | ����3��Һ�еμ�������ˮ | ��ɫ��ȥ | �ȱ�죬����ɫ |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ʵ����
ij�о���ѧϰС�飬Ϊ��̽���������Ƶ�ǿ�����ԣ��������ͼ��ʵ��װ�á�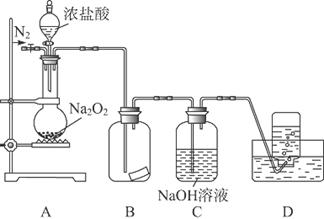
ʵ�鲽�輰�������£�
�ټ��װ�������Ժ�װ��ҩƷ������������
�ڻ���ͨ��һ������N2��װ��D���Ӻ�(����ĩ��δ���뼯��ƿ��)������Բ����ƿ�л����μ�Ũ���ᣬ��Ӧ���ң���������ɫ���塣
��һ��ʱ�������ĩ�����뼯��ƿ���ռ����塣װ��D���ռ�����ʹ�����ǵ�ľ����ȼ����ɫ���塣
�ܷ�Ӧ�����رշ�Һ©���Ļ�������ͨ��һ������N2����װ����������ɫ��
�ش��������⣺
��1��װ��B�е�ʪ��ĺ�ɫֽ����ɫ��֤��A�з�Ӧ��________(�ѧʽ)���ɡ���B�иķ�ʪ��ĵ���KI��ֽ����ƾ��ֽ������������֤���������ۣ��������ӷ���ʽ˵��ԭ��________________��
��2��װ��C��������_________________________________________________________��
��3����ͬѧ��ΪO2��Na2O2�������е�HCl��ԭ���á���ͬѧ��Ϊ�˽��۲���ȷ�������ܵ�����Ϊ��_________________________________________________________________��
��______ __��
��4��ʵ��֤����Na2O2��������HCl��Ӧ����ɲ���ƽ�û�ѧ����ʽ��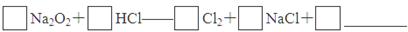
�÷�Ӧ________(��ܡ����ܡ�)����ʵ���ҿ�����ȡ������Cl2��������___________________________________________________________________________________________________________________________________(Ҫ����Ҫ��)��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ʵ����
ij�Ȼ�����Ʒ��������FeCl2���ʡ���Ҫ�ⶨ������Ԫ�ص�����������ʵ�鰴���²�����У�
������������̣��ش��������⣺
��1������I��������Һ�����õ��IJ����������ձ����������⣬�������� �� .�����������ƣ�
��2�����в�������ʹ������ҺŨ��ƫС����________________����д��ţ���
��δϴ���ձ��Ͳ�����
�ڶ���ʱ��������ƿ�Ŀ̶���
������Һǰ����ƿ������������ˮ
��ҡ�Ⱥ���Һ����ڿ̶��ߺ������ˮ����Һ����̶�������
��3����д��������ˮ���������ӷ���ʽ ��
��4����������Ƿ��Ѿ�ϴ�Ӹɾ��IJ����� ��
��5����ԭ��Ʒ����aΪ50g�����Ⱥ����ɫ��������bΪ3g������Ʒ����Ԫ�ص����������� ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ʵ����
��������������Ԫ�أ�����ȱ����ƶѪ�ij��������Ƿ��ò���ҩ��������ơ�(��Ҫ�ɷ֣��������������ʰ���ɫ)���г���һ�ֳ����IJ���ҩ���ҩƷ������ˮ�������������е�θ�ᡣ
ijͬѧΪ�˼�⡰�����ơ�ҩƬ��Fe2���Ĵ��ڣ���Ʋ�����������ʵ�飺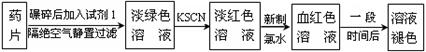
��1���Լ�1�� ������������ˮ����Һ�з��������ӷ�Ӧ����ʽ�ǣ� �� ��
��2������KSCN��Һ����δ��������ˮ������£���Һ��Ҳ�����˺�ɫ������ܵ�ԭ���� ��
��3����ʵ���з��ַ���һ��ʱ�䣬��Һ����ɫ������ȥ��Ϊ�˽�һ��̽����Һ��ɫ��ԭ�ס��ҡ�����λͬѧ���Ƚ����˲��룺
| ��� | �� �� |
| �� | ��Һ�еģ�3��Fe�ֱ���ԭΪ��2��Fe |
| �� | ��Һ�е�SCN������������ˮ���� |
| �� | ���Ƶ���ˮ����Ư���ԣ�������ҺƯ�� |
| ��� | ʵ����� | Ԥ������ͽ��� |
| �� | | |
| | | |
| | | |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ʵ����
Ϊ�ⶨNa2CO3��NaHCO3����������Na2CO3��������������ȡһ����������Ʒ����ͬѧ����ͼI��ʾװ�ò�������CO2���������ͬѧ����ͼII��ʾװ��ͨ������ܵ����ز�������CO2����������֪����ϡ�����������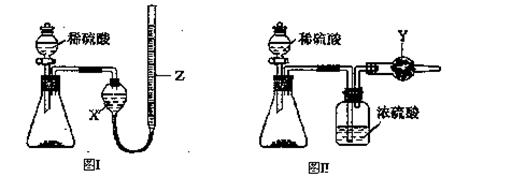
��l��ʢ��ϡ�������������Ϊ ��
��2���Լ�XΪ ���Լ�YΪ ��
��3����ͬѧ�ڽ���ʵ��ʱ��Ϊ��С��Ӧע��������У���ѡ����ĸ�� ��
A������ǰӦʹ����װ����ȴ������
B������Z�ĸ߶�ʹ����װ������Һ����ƽ
C������ʱ������Z�ڰ�Һ����͵�����
D������ǰӦͨ��һ������N2ʹ���ɵ�CO2ȫ����������װ��
��4������ͬѧ��ʵ�鷽������ʵ�飬ʹ��õ�Na2CO3����������ƫ�ߵ������У�дһ�֣�
��ʹ��õ�Na2CO3����������ƫ�͵�������
��дһ�֣� ��
��5��Ϊ�����ͬ�IJⶨ��������ʵ�鷽�����ܴﵽʵ��Ŀ�ĵ��� ����ѡ����ĸ����
A��ȡmg�����������Ba��OH��2��Һ��ַ�Ӧ�����ˡ�ϴ�ӡ���ɵ�ng����
B��ȡmg����������������ַ�Ӧ������Һ���ȡ����ɡ����յ�ng����
C��ȡmg������ּ��ȣ�������������ng
D����ͼIIװ���е�ϡ�����Ϊϡ�������ʵ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ʵ����
������һ����Ҫ�����ϣ���ѧʽΪFe2O3��xH2O���㷺����Ϳ�ϡ������ϡ��Ľ���Ʒ�ȹ�ҵ��ʵ����ģ�ҵ��������������Fe2O3��������CaO��MgO�ȣ��ͻ�����ۣ���Ҫ�ɷ�ΪFeS2���Ʊ����Ƶ��������£�
��1����������������ж��õ��������������������ֲ����е����÷ֱ��� �� ��
��2���Լ�a���ѡ�� ����ѡ��ʹ�õ��У����ۡ�������ŨHNO3������������ ��
��3���������������õ�����������װ�ÿ�����ʵ�����ư������� ������ţ���
��4��������ҺZ�к��� �ķ����� ��
�ķ����� ��
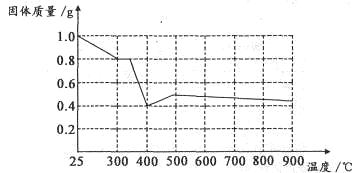
��5����������֪���ڲ�ͬ�¶���Fe2O3��CO��ԭ���������ΪFe3O4��FeO��Fe�����������뷴Ӧ�¶ȵĹ�ϵ����ͼ��ʾ��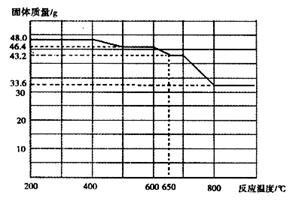
����ͼ���ƶ�670��ʱFe2O3��ԭ����Ļ�ѧʽΪ �������һ����ʵ�飬֤���û�ԭ����ijɷ֣�����ʵ�����������ͽ��ۣ��� ��������ѡ���ɹ�ѡ����Լ���ϡH2SO4��ϡ���ᡢH2O2��Һ��NaOH��Һ��KSCN��Һ��
�鿴�𰸺ͽ���>>
����ѧУ��ѡ - ��ϰ���б� - �����б�
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com