
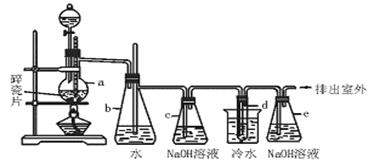
1��2![]() ��������������Ϳ����������Ӽ���������������ɫҺ�壬�ܶ�2.18g��cm3���е�131.4�棬�۵�9.79�棬������ˮ�������ڴ����ѡ���ͪ���л��ܼ�����ʵ�����п�������ͼ��ʾװ���Ʊ�1��2
��������������Ϳ����������Ӽ���������������ɫҺ�壬�ܶ�2.18g��cm3���е�131.4�棬�۵�9.79�棬������ˮ�������ڴ����ѡ���ͪ���л��ܼ�����ʵ�����п�������ͼ��ʾװ���Ʊ�1��2![]() �������飮���з�Һ������ƿa��װ���Ҵ���Ũ����Ļ��Һ���Թ�d��װ��Һ��(���渲������ˮ)��
�������飮���з�Һ������ƿa��װ���Ҵ���Ũ����Ļ��Һ���Թ�d��װ��Һ��(���渲������ˮ)��

��д���пհף�
(1)д���������Ʊ�1��2![]() ���������������ѧ����ʽ________
���������������ѧ����ʽ________
(2)��ȫƿb���Է�ֹ�����������Լ��ʵ�����ʱ�Թ�d�Ƿ�����������д����������ʱƿb�е�����________________��
(3)����c��NaOH��Һ������________________��
(4)ijѧ��������ʵ��ʱ��ʹ��һ������Һ�壬����ȫ����ɫʱ���������Ҵ���ŨH2SO4���Һ����������������³������࣮���װ�õ�������û�����⣬�Է�������ܵ�ԭ��_________��
|
����(1)CH3CH2OH ����CH2��CH2��Br2��CH2Br��CH2Br ����(2)b��ˮ����½����������е�ˮ��������������� ����(3)��ȥ��ϩ�д�������������(CO2��SO2)�� ����(4)����ϩ����(��ͨ��Һ��)�ٶȹ��죮 ������ʵ������У��Ҵ���ŨH2SO4�Ļ��Һû��Ѹ��������170�森 ���������� �������������Ѷ��ǡ���ȫƿ�����á�����ϸ�������Թ�d����ʱ��ʵ������ߵ������൱��ͨ����ϴƿ�����ɰ�bƿ�е�Һ�����뵽ֱ�ι��У� |


 ���ɶ��ȫ���ƿؾ�ϵ�д�
���ɶ��ȫ���ƿؾ�ϵ�д�
| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ��Ķ�����

| Ũ���� |
| 170�� |
| Ũ���� |
| 170�� |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
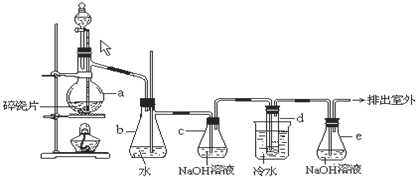 1��2-��������������Ϳ����������Ӽ���������������ɫҺ�壬�ܶ�2.18g?cm-3���е�131.4�棬�۵�9.79�棬������ˮ�������ڴ����ѡ���ͪ���л��ܼ�����ʵ�����п�������ͼ��ʾװ���Ʊ�1��2-�������飬���з�Һ©������ƿa��װ���Ҵ���Ũ����Ļ��Һ���Թ�d��װ��Һ�壨���渲������ˮ����
1��2-��������������Ϳ����������Ӽ���������������ɫҺ�壬�ܶ�2.18g?cm-3���е�131.4�棬�۵�9.79�棬������ˮ�������ڴ����ѡ���ͪ���л��ܼ�����ʵ�����п�������ͼ��ʾװ���Ʊ�1��2-�������飬���з�Һ©������ƿa��װ���Ҵ���Ũ����Ļ��Һ���Թ�d��װ��Һ�壨���渲������ˮ�����鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2011-2012ѧ������ʡ�����и�����ѧ�ڵ�һ���¿���ѧ�Ծ� ���ͣ�ʵ����
��9�֣�1��2 - ��������������Ϳ����������Ӽ���������������ɫҺ�壬�ܶ�2.18 g��cm-3���е�131.4�棬�۵�9.79�棬������ˮ�������ڴ����ѡ���ͪ���л��ܼ�����ʵ�����п�������ͼ��ʾװ���Ʊ�1��2- �������顣���з�Һ©������ƿa��װ���Ҵ���Ũ����Ļ��Һ���Թ�d��װ��Һ��(���渲������ˮ)����д���пհ�:

����(1)д���������Ʊ�1��2-���������������ѧ��Ӧ����ʽ��
����____________________________________________________________
����____________________________________________________________
(2)��ȫƿb���Է�ֹ�����������Լ��ʵ�����ʱ�Թ�d�Ƿ�����������д����������ʱƿb�е�����
_________________________________��
(3)����c��NaOH��Һ��������:__________________________________��
(4)ijѧ��������ʵ��ʱ��ʹ��һ������Һ�壬����ȫ����ɫʱ���������Ҵ���Ũ������Һ����������������³������ࡣ���װ�õ�������û�����⣬�Է�������ܵ�ԭ���ǣ�
����____________________________________________________________
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2012�����������ѧ�߶�3���¿���ѧ�Ծ� ���ͣ�ʵ����
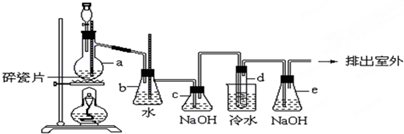
��8�֣�1��2����������������Ϳ����������Ӽ���������������ɫҺ�壬�ܶ�2.18 g��cm��3���е�131.4�棬�۵�9.79�棬������ˮ���������л��ܼ�����ʵ�����п�������ͼ��ʾװ���Ʊ�1��2���������顣���з�Һ©������ƿa��װ�оƾ���ŨH2SO4�Ļ��Һ���Թ�d��װ��Һ��(���渲������ˮ)����д���пհף�
(1)д��ʵ�����Ʊ�1��2�����������������ѧ��Ӧ����ʽ ��
��
(2)��ȫƿb���Է�ֹ�����������Լ��ʵ�����ʱ�Թ�d�Ƿ�����������д����������ʱƿb�е����� ��
(3)����c��NaOH��Һ��������__________________________________��
�鿴�𰸺ͽ���>>
����ѧУ��ѡ - ��ϰ���б� - �����б�
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com