
ʯī�ڲ�����������ҪӦ�á�ij����ʯī�к�SiO2(7.8%)��Al2O3(5.1%)��Fe2O3(3.1%)��MgO(0.5%)�����ʡ���Ƶ��ᴿ���ۺ����ù������£�
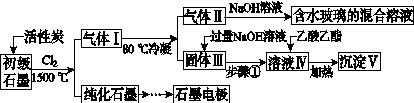
(ע��SiCl4�ķе�Ϊ57.6 �棬�����Ȼ���ķе������150 ��)
(1)��Ӧ����ͨ��Cl2ǰ����ͨһ��ʱ��N2����ҪĿ����____________________��
(2)���·�Ӧ��ʯī�����������ʾ�ת��Ϊ��Ӧ���Ȼ��������е�̼��������ҪΪ________�����������ij��õ�ˮ�����Ļ�ѧ��Ӧ����ʽΪ____________________________________________��
(3)�����Ϊ�����衢________��������Һ���е���������________��
(4)����Һ�����ɳ��������ܷ�Ӧ�����ӷ���ʽΪ______________________________________________��100 kg����ʯī�����ܻ�â�������Ϊ______kg��
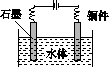
(5)ʯī��������Ȼˮ����ͭ���ĵ绯ѧ�����������ͼ����ʾ��ͼ��������Ӧ��ע��
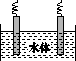
(1)�ų�װ���еĿ��� ��
(2)CO�� SiCl4��6NaOH===Na2SiO3��4NaCl��3H2O ��
(3)���ˡ�AlO ��Cl�� ��
��Cl�� ��
(4)AlO ��CH3COOCH2CH3��2H2O
��CH3COOCH2CH3��2H2O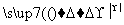 CH3COO����CH3CH2OH��Al(OH)3����7.8 ��
CH3COO����CH3CH2OH��Al(OH)3����7.8 ��
(5)
[����] (1)ͨ��N2��Ŀ����Ϊ���ų�װ���еĿ���(�ر�������)����ֹ�ڸ���ʱʯī��������Ӧ��(2)���·�Ӧ��SiO2��Al2O3��Fe2O3��MgO�ֱ�ת��ΪSiCl4��AlCl3��FeCl3��MgCl2����Ϊʯī�ǹ����ģ��ʸ��������£�C��SiO2��Fe2O3��Ӧ���ɵ���CO������SiCl4�ķе�Ϊ57.6 �棬����80 �棬�������ΪSiCl4����NaOH��Һ��ˮ������Na2SiO3��NaCl: SiCl4��6NaOH===Na2SiO3��4NaCl��3H2O��(3)AlCl3��FeCl3��MgCl2�ķе������150 �棬��80 ���±�Ϊ����� AlCl3��FeCl3��MgCl2����NaOH��Ӧ������NaAlO2��Fe(OH)3��Mg(OH)2��NaCl��ͨ�����˽�����Fe(OH)3��Mg(OH)2�˳����õ�����Һ����Ҫ��NaAlO2��NaCl��(4)NaAlO2����ˮ�����Һ�Լ��ԣ�NaAlO2��2H2O Al(OH)3��NaOH������������������ˮ�⣺CH3COOCH2CH3��NaOH
Al(OH)3��NaOH������������������ˮ�⣺CH3COOCH2CH3��NaOH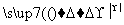 CH3COONa��CH3CH2OH����ʹNaAlO2����ˮ������Al(OH)3��������Һ��ת��Ϊ�������ķ�ӦΪNaAlO2��2H2O��CH3COOCH2CH3
CH3COONa��CH3CH2OH����ʹNaAlO2����ˮ������Al(OH)3��������Һ��ת��Ϊ�������ķ�ӦΪNaAlO2��2H2O��CH3COOCH2CH3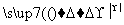 Al(OH)3����CH3COONa��CH3CH2OH������Alԭ���غ㣬��֪100 kg����ʯī�ɵ�m[Al(OH)3]��
Al(OH)3����CH3COONa��CH3CH2OH������Alԭ���غ㣬��֪100 kg����ʯī�ɵ�m[Al(OH)3]��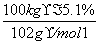 ��2��78 g��mol��1��7.8 kg��
��2��78 g��mol��1��7.8 kg��
(5)ˮ����ͭ���ĵ绯ѧ���������˵�Ᵽ�������÷�����ʯī��������ͭ��������������ӵ���������������������ʯī��ͭ��ֱ���������γ�ԭ��أ���ͭ�����������������Բ��ɲ��á�


 ��У����ϵ�д�
��У����ϵ�д�
| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
mgij����M�뺬n mol HCl������ǡ����ȫ��Ӧ������MCl2��H2����ý��������ԭ������Ϊ�� ��
A��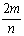 B��
B��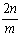 C��
C��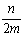 D��
D��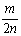
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
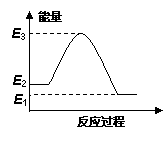
���淴Ӧ��A(g)  B(g)���䷴Ӧ�����������仯����ͼ��ʾ������˵���������
B(g)���䷴Ӧ�����������仯����ͼ��ʾ������˵���������
����Ea��ʾ��ܣ�Ea(��)��Ea(��)�ֱ��ʾ�����淴Ӧ��ܣ�
A���÷�Ӧ�� ��H = Ea(��) �� Ea(��)
B���÷�Ӧ��Ea(��) = E3 �� E2
C��ֻҪ�¶����ߣ��ÿ��淴Ӧ�ͻ����淴Ӧ�������
D�����淴ӦB(g)  A(g)����H = E2 �� E1
A(g)����H = E2 �� E1
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
| W | Y | R | |
| Z |
���ΪԪ�����ڱ�ǰ�����ڵ�һ���֣������й�X��W��Y��R��Z����Ԫ�ص������У���ȷ����
A�����³�ѹ�£�����Ԫ�ض�Ӧ�ĵ���������������̬
B��YԪ������������Ӧˮ��������Ա�WԪ�ص���
C��Y��Z�������ӵ��Ӳ�ṹ����Rԭ�ӵ���ͬ
D��W���⻯���X���⻯���ȶ�
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
���й�������Ӧ�ú���ɵ�˵����ȷ����(����)
A��P2O5�����ڸ���Cl2��NH3
B������ȼ������Ҫ�ɷ��Ǽ����ˮ
C��CCl4�����ڼ�����ˮ�͵�ˮ
D��Si��SiO2������������ά
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
Ǧ���仯������������ء������豸��X���߷������ϵȡ��ش��������⣺
(1)Ǧ��̼��ͬ��Ԫ�أ���̼��4�����Ӳ㡣Ǧ��Ԫ�����ڱ���λ��Ϊ��________���ڡ���________�壻PbO2�����Ա�CO2������________(�ǿ��������)��
(2)PbO2��Ũ���Ṳ�����ɻ���ɫ���壬��Ӧ�Ļ�ѧ����ʽΪ_______________________��
(3)PbO2����PbO�����������Һ��Ӧ�Ƶã���Ӧ�����ӷ���ʽΪ___________________��PbO2Ҳ����ͨ��ʯīΪ�缫��Pb(NO3)2��Cu(NO3)2�Ļ����ҺΪ���Һ�����ȡ�����������ĵ缫��ӦʽΪ____________________�������Ϲ۲쵽��������____________________�������Һ�в�����Cu(NO3)2�����������ĵ缫��ӦʽΪ______________________________������������Ҫȱ����____________________��
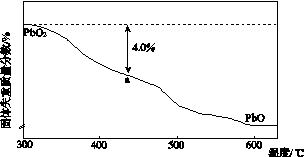
(4)PbO2�ڼ��ȹ��̷����ֽ��ʧ����������ͼ��ʾ����֪ʧ�������ϵ�a��Ϊ��Ʒʧ��4.0%(��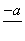 ��100%)�IJ������塣��a�������ɱ�ʾΪPbOx��mPbO2��nPbO����ʽ����xֵ��m��nֵ_______________________________________
��100%)�IJ������塣��a�������ɱ�ʾΪPbOx��mPbO2��nPbO����ʽ����xֵ��m��nֵ_______________________________________
________________________________________________________________________
________________________________________________________________________��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
ʵ���ҴӺ����Һ(��H2O�⣬����CCl4��I2��I����)�л��յ⣬��ʵ��������£�

(1)���Һ�м����Թ�����Na2SO3��Һ������Һ�е�I2��ԭΪI���������ӷ���ʽΪ__________________���ò�����I2��ԭΪI����Ŀ����______________________��
(2)����X������Ϊ________��
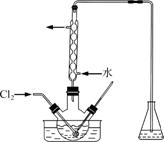
(3)����ʱ����������ƿ�н���I����ˮ��Һ���������pHԼΪ2������ͨ��Cl2����40 �����ҷ�Ӧ(ʵ��װ����ͼ��ʾ)��
ʵ������ڽϵ��¶��½��е�ԭ����______________����ƿ��ʢ�ŵ���ҺΪ________��
(4)��֪��5SO ��2IO
��2IO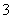 ��2H��===I2��5SO
��2H��===I2��5SO ��H2O
��H2O
ij�����ˮ(pHԼΪ8)��һ������I2�����ܴ���I����IO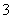 �е�һ�ֻ����֡��벹���������麬���ˮ���Ƿ���I����IO
�е�һ�ֻ����֡��벹���������麬���ˮ���Ƿ���I����IO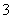 ��ʵ�鷽����ȡ���������ˮ��CCl4�����ȡ����Һ��ֱ��ˮ���õ�����Һ���鲻���еⵥ�ʴ��ڣ�________________________________________________________________________
��ʵ�鷽����ȡ���������ˮ��CCl4�����ȡ����Һ��ֱ��ˮ���õ�����Һ���鲻���еⵥ�ʴ��ڣ�________________________________________________________________________
________________________________________________________________________
________________________________________________________________________��
ʵ���пɹ�ѡ����Լ���ϡ���ᡢ������Һ��FeCl3��Һ��Na2SO3��Һ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
���ʯ��ʯī����̼�ĵ��ʣ�ʯī��һ�������¿���ת��Ϊ���ʯ����֪12gʯī��ȫת��Ϊ���ʯʱ��Ҫ����E kJ������������˵����ȷ���ǣ� ��
A.���ʯ��ʯī��Ϊͬλ�� B.ʯī������ʯ�ȶ�
C.���ʯ����ʯī�ȶ� D.��������ʯī����ʯ��ȫȼ�գ�ʯī�ų���������
�鿴�𰸺ͽ���>>
����ѧУ��ѡ - ��ϰ���б� - �����б�
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com