����⣺��1��C�����������ն�����̼����֤��ˮ��������õ��������������
�ʴ�Ϊ��NaOH��Һ��
��2��A����Ƥ�ܿ��Թ�ͨ��ƿ�ͷ�Һ©�������ѹ���ã�ʹ���������£����ҿ��Ա��������ſ���ʹ�������ƫ��
�ʴ�Ϊ�����ѹ���ã�ʹ���������£����������ſ���ʹ�������ƫ��
��3��a��װ��A��B�еĿ����ᵼ�²�õ��������ƫ�ߣ��Բⶨ�������Ӱ�죬��a��ȷ��
b���ڵ���U���������е�Һ����ƽ�Ĺ����У��ų���װ��C�еĿ����Բⶨ���������Ӱ�죬��b����
c������ʱU���������е�Һ��Ҫ������ƽ������ҵͣ�����Na
2O
2�Ĵ��Ȼ�ƫ��c��ȷ��
d������ʱU���������е�Һ������Ҹ�ʱ������Na
2O
2�Ĵ��Ȼ�ƫС����d����
��ѡac��
��4��ͼ�����õ���������Һ�ܣ�����Һ�ܷ��������Һ�������У�ʹ���ڼ�˿��������ڱڣ���������б����Һ���ִ�ֱ���ſ�ʳָ��ʹ��Һ�������ڱ���Ȼ���£�����Һ������Һ�������ٵȴ�15�룬ȡ����Һ�ܣ����ڹܿڵ�����Һ�岻Ҫ������
�ʴ��ǣ�D����Һ�ܣ�
��5�����εζ����ı�Һ���������Ч�ģ��������ĵı�Һ�����ƽ������ǣ�
mL=25mL���������ơ������ƺ����ᷴӦ�Ĺ�ϵʽ�ǣ�Na
2O
2��2HCl��Na
2O��2HCl���������ƺ��������ܵ����ʵ����ǣ�n��Na
2O
2��=
n��HCl��=0.5��0.025L��0.1000mol?L
-1=0.00125mol��1000.00mL��Һ��Һ�к��е������ƺ��������ܵ����ʵ����ǣ�0.00125��
=0.05mol��
���������xmol�������������ʵ����ǣ�0.05-x��mol��
����������ϵ�ɵã�78x+62����0.05-x��=3.500��
���x=0.025mol���������Ƶ����������ǣ�
��100%=55.7%��
�ʴ��ǣ�55.7%��

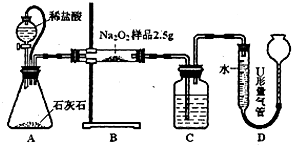 Ϊ�ⶨ����Na2O���ʵ�Na2O2��Ʒ�Ĵ��ȣ��ס��Ҷ�λͬѧ����˶��ֲ�ͬ��ʵ�鷽����
Ϊ�ⶨ����Na2O���ʵ�Na2O2��Ʒ�Ĵ��ȣ��ס��Ҷ�λͬѧ����˶��ֲ�ͬ��ʵ�鷽����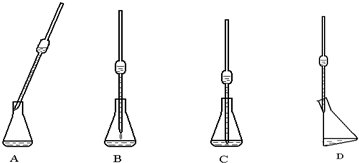



 ���أ�
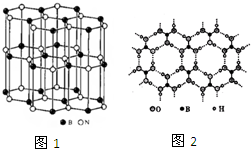
���أ� ������ɰ��Na2B4O7���ڸ��¸�ѹ�·�Ӧ���Ի�������
������ɰ��Na2B4O7���ڸ��¸�ѹ�·�Ӧ���Ի�������