
��14�֣���������;�㷺����Ҫ��������Ӳ�ʻ����µĺϽ��Լ����ݵĵ�˿�������£����ܱ���������H2��ԭWO3�ɵõ������٣����ܷ�ӦΪ��
WO3 (s) + 3H2 (g)  W (s) + 3H2O (g)
W (s) + 3H2O (g)
��ش��������⣺
�� ������Ӧ�Ļ�ѧƽ�ⳣ������ʽΪ___________________________��
�� ij�¶��·�Ӧ��ƽ��ʱ��H2��ˮ�����������Ϊ2:3����H2��ƽ��ת����Ϊ_____________________�����¶ȵ����ߣ�H2��ˮ����������ȼ�С����÷�ӦΪ___________��Ӧ������ȡ����ȡ�����
�������ܷ�Ӧ���̴��·�Ϊ�����Σ�������Ҫ�ɷ����¶ȵĹ�ϵ���±���ʾ��
|
�¶� |
25�� ~ 550�� ~ 600�� ~ 700�� |
|
��Ҫ�ɷ� |
WO3 W2O5 WO2 W |
��һ�η�Ӧ�Ļ�ѧ����ʽΪ___________________________��580��ʱ���������ʵ���Ҫ�ɷ�Ϊ________������WO3��ȫת��ΪW��������������H2���ʵ���֮��Ϊ____________________________________��
�� ��֪���¶ȹ���ʱ��WO2 (s)ת��ΪWO2 (g)��
WO2 (s) + 2H2 (g)  W (s) + 2H2O (g)
��H �� +66.0 kJ��mol��1
W (s) + 2H2O (g)
��H �� +66.0 kJ��mol��1
WO2 (g) + 2H2(g)  W (s) + 2H2O (g)
��H �� ��137.9 kJ��mol��1
W (s) + 2H2O (g)
��H �� ��137.9 kJ��mol��1
��WO2 (s)  WO2 (g) �Ħ�H ��
______________________��
WO2 (g) �Ħ�H ��
______________________��
�� ��˿�ƹ��е�W��ʹ�ù����л����ӷ���ʹ��˿��ϸ������I2���ӳ��ƹܵ�ʹ���������乤��ԭ��Ϊ��W (s) +2I2 (g) 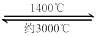 WI4 (g)������˵����ȷ����________________��
WI4 (g)������˵����ȷ����________________��
a���ƹ��ڵ�I2��ѭ��ʹ��
b��WI4�ڵ�˿�Ϸֽ⣬������W�ֳ����ڵ�˿��
c��WI4�ڵƹܱ��Ϸֽ⣬ʹ�ƹܵ������ӳ�
 d���¶�����ʱ��WI4�ķֽ����ʼӿ죬W��I2�Ļ������ʼ���
d���¶�����ʱ��WI4�ķֽ����ʼӿ죬W��I2�Ļ������ʼ���
����1��k=
����2��60%�����ȣ���3��2WO3+H2 W2O5+H2O��W2O5��WO2��1:1:4����4��+203.9kJ��mol-1����5��ab��
W2O5+H2O��W2O5��WO2��1:1:4����4��+203.9kJ��mol-1����5��ab��
 ������������1����������Ļ�ѧ����ʽ�����е�WO3��W��Ϊ���壬ƽ�ⳣ���в�д�룬��k= ����2���ɷ���ʽ��WO3+3H2
������������1����������Ļ�ѧ����ʽ�����е�WO3��W��Ϊ���壬ƽ�ⳣ���в�д�룬��k= ����2���ɷ���ʽ��WO3+3H2 W+3H2O�������ʵĻ�ѧ��������ͬ����֪ƽ��ʱ���ߵ������Ϊ2:3����������ƽ��ת����Ϊ����3/5����100%=60%���¶�����ʱ��������ˮ����������ȼ�С��˵��ƽ���������ƶ���˵���÷�Ӧ�������ȣ���3�����ݱ����и����ķ�Ӧ�������Σ���һ�Σ�WO3ת��ΪW2O5������ʽ�ɱ�ʾΪ��2WO3+H2
W+3H2O�������ʵĻ�ѧ��������ͬ����֪ƽ��ʱ���ߵ������Ϊ2:3����������ƽ��ת����Ϊ����3/5����100%=60%���¶�����ʱ��������ˮ����������ȼ�С��˵��ƽ���������ƶ���˵���÷�Ӧ�������ȣ���3�����ݱ����и����ķ�Ӧ�������Σ���һ�Σ�WO3ת��ΪW2O5������ʽ�ɱ�ʾΪ��2WO3+H2 W2O5+H2O����580��ʱ����Ӧ����ڶ��Σ��������ʵ���Ҫ�ɷ��ǣ�W2O5��WO2���ڶ��εķ���ʽ�ɱ�ʾΪ��W2O5+H2
W2O5+H2O����580��ʱ����Ӧ����ڶ��Σ��������ʵ���Ҫ�ɷ��ǣ�W2O5��WO2���ڶ��εķ���ʽ�ɱ�ʾΪ��W2O5+H2 2WO2+H2O�������εķ���ʽΪ�� WO2+2H2
2WO2+H2O�������εķ���ʽΪ�� WO2+2H2 W+2H2O�����������ĵ����������ʵ���Ϊ��1:1:4����4����������������Ȼ�ѧ����ʽ��WO2(s)+2H2(g)
W+2H2O�����������ĵ����������ʵ���Ϊ��1:1:4����4����������������Ȼ�ѧ����ʽ��WO2(s)+2H2(g) W(s)+2H2O(g)
��H= +66.0 kJ��mol-1�٣�
WO2(g)+2H2(g)
W(s)+2H2O(g)
��H= +66.0 kJ��mol-1�٣�
WO2(g)+2H2(g) W(s)+2H2O(g)
��H=-137.9kJ��mol-1�ڣ�����Ӧ��-�ڵõ���WO2(s)
W(s)+2H2O(g)
��H=-137.9kJ��mol-1�ڣ�����Ӧ��-�ڵõ���WO2(s)  WO2(g) ��H= +203.9kJ��mol-1����5��������˿�ƹܵķ�Ӧԭ������֪�ƹ��ڵĵⵥ�ʿ�ѭ��ʹ�ã�a�ԣ�����ⵥ�ʵ������Ǽ�����˿��ϸ����b�ԣ������ĵ⻯������1400�����ɣ�����3000��ʱ�ֽ⣬��Ӧ�ڵ�˿���γɣ�c�����¶�����ʱ�������Ƿֽ����ʺͻ������ʶ��ӿ죬d����
WO2(g) ��H= +203.9kJ��mol-1����5��������˿�ƹܵķ�Ӧԭ������֪�ƹ��ڵĵⵥ�ʿ�ѭ��ʹ�ã�a�ԣ�����ⵥ�ʵ������Ǽ�����˿��ϸ����b�ԣ������ĵ⻯������1400�����ɣ�����3000��ʱ�ֽ⣬��Ӧ�ڵ�˿���γɣ�c�����¶�����ʱ�������Ƿֽ����ʺͻ������ʶ��ӿ죬d����
���㶨λ������Ϊ�Խ����ٵ���ȡΪ�������ۺϿ����˻�ѧƽ�ⳣ����ƽ��ת���ʡ���ѧƽ����ƶ�����˹���ɡ���ѧ����ʽ����д��֪ʶ��



| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
| ���� |
| c3(H2O) |
| c3(H2) |
| c3(H2O) |
| c3(H2) |
| 1400�� |
| Լ3000�� |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ��Ķ�����
| ���� |
| c3(H2O) |
| c3(H2) |
| c3(H2O) |
| c3(H2) |
| �¶� | 25�桫550�桫600�桫700�� |
| ��Ҫ�ɷ� | WO3 W2O5 WO2 W |
| ||
| ||
| ||
| Լ3000�� |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2012����ͨ�ߵ�ѧУ����ȫ��ͳһ�������ۻ�ѧ���֣������������� ���ͣ������
��14�֣���������;�㷺����Ҫ��������Ӳ�ʻ����µĺϽ��Լ����ݵĵ�˿�������£����ܱ���������H2��ԭWO3�ɵõ������٣����ܷ�ӦΪ��
WO3 (s) + 3H2 (g)  W (s) + 3H2O (g)
W (s) + 3H2O (g)
��ش��������⣺
�� ������Ӧ�Ļ�ѧƽ�ⳣ������ʽΪ___________________________��
�� ij�¶��·�Ӧ��ƽ��ʱ��H2��ˮ�����������Ϊ2:3����H2��ƽ��ת����Ϊ_____________________�����¶ȵ����ߣ�H2��ˮ����������ȼ�С����÷�ӦΪ___________��Ӧ������ȡ����ȡ�����
�������ܷ�Ӧ���̴��·�Ϊ�����Σ�������Ҫ�ɷ����¶ȵĹ�ϵ���±���ʾ��
| �¶� | 25�� ~ 550�� ~ 600�� ~ 700�� |
| ��Ҫ�ɷ� | WO3 W2O5 WO2 W |
 W (s) + 2H2O (g) ��H�� +66.0 kJ��mol��1
W (s) + 2H2O (g) ��H�� +66.0 kJ��mol��1 W (s) + 2H2O (g) ��H �� ��137.9 kJ��mol��1
W (s) + 2H2O (g) ��H �� ��137.9 kJ��mol��1 WO2 (g) �Ħ�H �� ______________________��
WO2 (g) �Ħ�H �� ______________________�� WI4 (g)������˵����ȷ����________________��
WI4 (g)������˵����ȷ����________________���鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2012-2013ѧ��㶫ʡʵ����ѧ�߶���ѧ�����л�ѧ�Ծ����������� ���ͣ������
��16�֣���������;�㷺����Ҫ��������Ӳ�ʻ����µĺϽ��Լ����ݵĵ�˿�������£����ܱ���������H2��ԭWO3�ɵõ������٣����ܷ�ӦΪ��
WO3��s��+3 H2��g�� W��s��+3 H2O��g��
W��s��+3 H2O��g��
��ش��������⣺
��1��������Ӧ�Ļ�ѧƽ�ⳣ������ʽΪ
��2��ij�¶��·�Ӧ��ƽ��ʱ�����¶ȵ����ߣ� H2��ˮ����������ȼ�С����÷�ӦΪ_____________��Ӧ(�� ���Ȼ����)
��3�������ܷ�Ӧ���̴��·�Ϊ�����Σ�������Ҫ�ɷ����¶ȵĹ�ϵ���±���ʾ��
| �¶� | 25 �� ~ 550 �� ~ 600�� ~ 700�� |
| ��Ҫ�ɷ� | WO3 W2O5 WO2 W |
 W��s��+2H2O��g�� ��H��+66 kJ·mol-1
W��s��+2H2O��g�� ��H��+66 kJ·mol-1 W��s��+2H2O��g�� ��H�� ��137.9 kJ·mol-1
W��s��+2H2O��g�� ��H�� ��137.9 kJ·mol-1 WO2��g���Ħ�H��_____ _______.
WO2��g���Ħ�H��_____ _______.
�鿴�𰸺ͽ���>>
����ѧУ��ѡ - ��ϰ���б� - �����б�
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com