
| 3 |
| 2 |
| 3 |
| 2 |
| 3 |
| 2 |
| 1 |
| 2 |
| 1 |
| 8 |


 ��������ϵ�д�
��������ϵ�д� ����˼ά����ѵ����ʱ��ѧ��ϵ�д�
����˼ά����ѵ����ʱ��ѧ��ϵ�д�
| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ��Ķ�����
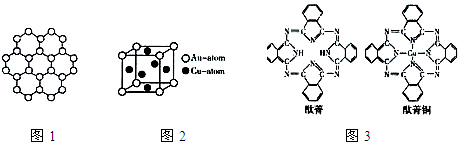
 ���ʼҿ�ѧԺ��������2010��ŵ��������ѧ������ Ӣ������˹�ش�ѧ��ѧ�Ұ�����?��ķ�Ϳ�˹̹��?ŵ��Ф����Ա���������ʯīϩ���Ϸ����Խ�о���ʯīϩ����̼ԭ�ӹ��ɵĵ���Ƭ״�ṹ���²��ϣ�����ԭ�Ӽ��ĺ�ȡ�����ĵ�ѧ���ܡ���ɫ�Ļ�ѧ�ȶ��Ժ�����ѧ�ȶ��ԣ��Ʊ�ʯīϩ������ʯī���뷨����ѧ����������ȣ�ʯīϩ�����ģ�ͼ����ӽṹ��ͼ1��ʾ��
���ʼҿ�ѧԺ��������2010��ŵ��������ѧ������ Ӣ������˹�ش�ѧ��ѧ�Ұ�����?��ķ�Ϳ�˹̹��?ŵ��Ф����Ա���������ʯīϩ���Ϸ����Խ�о���ʯīϩ����̼ԭ�ӹ��ɵĵ���Ƭ״�ṹ���²��ϣ�����ԭ�Ӽ��ĺ�ȡ�����ĵ�ѧ���ܡ���ɫ�Ļ�ѧ�ȶ��Ժ�����ѧ�ȶ��ԣ��Ʊ�ʯīϩ������ʯī���뷨����ѧ����������ȣ�ʯīϩ�����ģ�ͼ����ӽṹ��ͼ1��ʾ���鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
 ��2012?��������ģ���������г�������X��ʯī�Ͷ������ѣ�TiO2����������ϣ������·�Ӧ�õ��Ļ������������Ԫ����ɣ��Ҷ��������մɲ��ϣ��ڻ���͵���������ҪӦ�ã��������������������գ�
��2012?��������ģ���������г�������X��ʯī�Ͷ������ѣ�TiO2����������ϣ������·�Ӧ�õ��Ļ������������Ԫ����ɣ��Ҷ��������մɲ��ϣ��ڻ���͵���������ҪӦ�ã��������������������գ�

| ||
| ||
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
 2010��10��5�ձ���ʱ��17ʱ45�����ʼҿ�ѧԺ��������2010��ŵ��������ѧ������Ӣ������˹�ش�ѧ��ѧ�Ұ�����?��ķ�Ϳ�˹̹��?ŵ��Ф����¶���Ķ�����ͻ���Եء���˺�ѵķ����ɹ���ó�������ʯīϩ����ͼ����������˵����ȷ���ǣ�������
2010��10��5�ձ���ʱ��17ʱ45�����ʼҿ�ѧԺ��������2010��ŵ��������ѧ������Ӣ������˹�ش�ѧ��ѧ�Ұ�����?��ķ�Ϳ�˹̹��?ŵ��Ф����¶���Ķ�����ͻ���Եء���˺�ѵķ����ɹ���ó�������ʯīϩ����ͼ����������˵����ȷ���ǣ��������鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
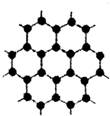 2010��10��5��17ʱ45�֣����ʼҿ�ѧԺ��������2010��ŵ��������ѧ������Ӣ������˹�ش�ѧ��ѧ�Ұ�����?��ķ�Ϳ�˹̹��?ŵ��Ф����Ա���������ʯīϩ���Ϸ����Խ�о���ʯīϩ��Ŀǰ�Ƽ��о����ȵ㣬�ɿ�����ʯī�IJ�״�ṹһ��һ��İ����õ��ĵ���̼ԭ�ӣ��ṹ��ͼ��ʾ�������������뵽ʯīϩ�п�����һ�־���ͻ���Ե��²���ʯī�飬����˵������ȷ���ǣ�������
2010��10��5��17ʱ45�֣����ʼҿ�ѧԺ��������2010��ŵ��������ѧ������Ӣ������˹�ش�ѧ��ѧ�Ұ�����?��ķ�Ϳ�˹̹��?ŵ��Ф����Ա���������ʯīϩ���Ϸ����Խ�о���ʯīϩ��Ŀǰ�Ƽ��о����ȵ㣬�ɿ�����ʯī�IJ�״�ṹһ��һ��İ����õ��ĵ���̼ԭ�ӣ��ṹ��ͼ��ʾ�������������뵽ʯīϩ�п�����һ�־���ͻ���Ե��²���ʯī�飬����˵������ȷ���ǣ�������| A��ʯīϩ�Ǹ߷��ӻ����� | B��ʯīϩ��ʯī�黥Ϊͬ�������� | C��һ��������ʯīϩ����H2�����ӳɷ�Ӧ | D�����ݽṹʾ��ͼ��֪��ʯīϩ���ܵ��� |
�鿴�𰸺ͽ���>>
����ѧУ��ѡ - ��ϰ���б� - �����б�
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com