
(17��)
![]() ij�о���С�����A-D������װ������й�ʵ��
ij�о���С�����A-D������װ������й�ʵ��
![]()
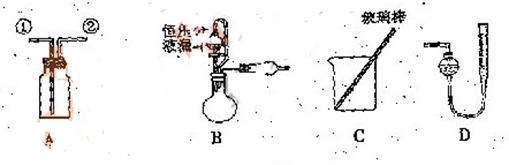
![]() ��ʵ��һ���ռ�NO���塣
��ʵ��һ���ռ�NO���塣
��1������ ![]() ��װ��A�ռ�NO���壬��ȷ�IJ����������� ������ţ���
��װ��A�ռ�NO���壬��ȷ�IJ����������� ������ţ���
![]() a.�Ӣٿڽ���������ˮ�������� ��������������b.�Ӣٿڽ�����������������
a.�Ӣٿڽ���������ˮ�������� ��������������b.�Ӣٿڽ�����������������
![]() c.�Ӣڿڽ���������ˮ���������������������� d..�Ӣڿڽ�����������������
c.�Ӣڿڽ���������ˮ���������������������� d..�Ӣڿڽ�����������������
![]() ��ʵ�����Ϊ��̽����п�������ϵ�п����������
��ʵ�����Ϊ��̽����п�������ϵ�п����������![]() �ͶƲ��ȣ���ѯ��֪п�����ڼZn+2NaOH=Na2ZnO3+H2���ݴˣ���ȡ���ΪS��˫���п���������������顢�Ƶ�����Ϊm1 g���ù����ռ��ˮ���Լ����������ʵ�鷽�����������ʵ�顣
�ͶƲ��ȣ���ѯ��֪п�����ڼZn+2NaOH=Na2ZnO3+H2���ݴˣ���ȡ���ΪS��˫���п���������������顢�Ƶ�����Ϊm1 g���ù����ռ��ˮ���Լ����������ʵ�鷽�����������ʵ�顣
![]() �����ף�ͨ������������Ӧ���ɵ����������ʵ��̽��ľĿ�ꡣ
�����ף�ͨ������������Ӧ���ɵ����������ʵ��̽��ľĿ�ꡣ
![]() ��2��ѡ��B���������� ����������ţ�����װ�ý���ʵ�顣
��2��ѡ��B���������� ����������ţ�����װ�ý���ʵ�顣
![]() ��3����ó�ַ�Ӧ���������������ΪVL����״������
��3����ó�ַ�Ӧ���������������ΪVL����״������![]() =������ ��
=������ ��
![]() ��4������Ʋ��ȣ�����Ҫ������һ������������������������ ��
��4������Ʋ��ȣ�����Ҫ������һ������������������������ ��
![]() ��5����װ��B�еĺ�ѹ��Һ©����Ϊ��ͨ��Һ©����������������ƫ����ƫС������Ӱ�족����
��5����װ��B�еĺ�ѹ��Һ©����Ϊ��ͨ��Һ©����������������ƫ����ƫС������Ӱ�족����
![]() �����ң�ͨ������������Ӧǰ�������ʵ��̽��Ŀ�ꡣѡ������C��ʵ�飬��������ַ�Ӧ���˳������ϴ�ӡ���ɣ��Ƶ�������Ϊm2g ��
�����ң�ͨ������������Ӧǰ�������ʵ��̽��Ŀ�ꡣѡ������C��ʵ�飬��������ַ�Ӧ���˳������ϴ�ӡ���ɣ��Ƶ�������Ϊm2g ��
![]() ��6��
��6��![]() ���������� ��
���������� ��
![]() ��������ͨ������������Ӧǰ���������������Լ��������������ֵ��ΪH2��������ʵ��̽��Ŀ�ꡣʵ��ͬ��ʹ������C��
��������ͨ������������Ӧǰ���������������Լ��������������ֵ��ΪH2��������ʵ��̽��Ŀ�ꡣʵ��ͬ��ʹ������C��
![]() ��7����ʵ�����Ƕȷ������������������� �����ң�����ڡ��������ڡ���ͬ�ڡ�����
��7����ʵ�����Ƕȷ������������������� �����ң�����ڡ��������ڡ���ͬ�ڡ�����
��(1)C (2)D (3) ![]() (��
(��![]() �����������𰸣�
�����������𰸣�
��4������п���ܶȣ������������𰸣�
��5��ƫ��
��6��![]() �������������𰸣�
�������������𰸣�
��7������
�����������⿼��ʵ���̽�����漰NO������ռ���������н��������ļ���ȡ���1��NO�������������������ʲ������ſ������ռ���ֻ������ˮ���ռ�����ˮʱӦ�ö̽���������2�������ף�Zn��Fe��ֻ��Zn������NaOH�������壬ͨ����ˮ�ռ����壬���ݷ�Ӧ����ʽ�������Zn������������������Ҫ��װ���в�H2�������Dװ�á���3��Zn��H2֮����Ϊ1��1����n(Zn)=V/22.4 mol,w (Zn)=m(Zn)/m1 = ![]() ����4������Zn��������������ܶȣ���������Zn�����������Zn�Ľ�������������Zn�ĸ߶ȣ���ȣ�����5����ѹʽ��Һ©�������������в��ֲ����ڷ�Һ©���Ϸ���������ʱ�ռ����ˣ���������ͨ©��ʱ�ռ���H2��һЩ����������Zn����ƫ��6�����ٵ�������ΪZn����������7������������H2��������ֵ����Ȼ������Ϊ������H2������С������ƫ���
����4������Zn��������������ܶȣ���������Zn�����������Zn�Ľ�������������Zn�ĸ߶ȣ���ȣ�����5����ѹʽ��Һ©�������������в��ֲ����ڷ�Һ©���Ϸ���������ʱ�ռ����ˣ���������ͨ©��ʱ�ռ���H2��һЩ����������Zn����ƫ��6�����ٵ�������ΪZn����������7������������H2��������ֵ����Ȼ������Ϊ������H2������С������ƫ���



| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
(17��)ij�о���С�����A-D������װ������й�ʵ��
![]()
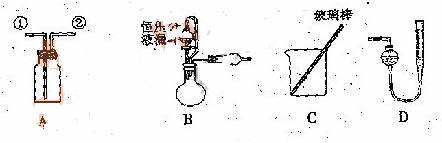
![]() ��ʵ��һ���ռ�NO���塣
��ʵ��һ���ռ�NO���塣
��1�� ![]() ��װ��A�ռ�NO���壬��ȷ�IJ����� ������ţ���
��װ��A�ռ�NO���壬��ȷ�IJ����� ������ţ���
![]() a.�Ӣٿڽ���������ˮ������ b.�Ӣٿڽ�����������������
a.�Ӣٿڽ���������ˮ������ b.�Ӣٿڽ�����������������
![]() c.�Ӣڿڽ���������ˮ������ d..�Ӣڿڽ�����������������
c.�Ӣڿڽ���������ˮ������ d..�Ӣڿڽ�����������������
![]() ��ʵ�����Ϊ��̽����п�������ϵ�п����������
��ʵ�����Ϊ��̽����п�������ϵ�п����������![]() �ͶƲ��ȣ���ѯ��֪п�����ڼZn+2NaOH=Na2ZnO3+H2���ݴˣ���ȡ���ΪS��˫���п���������������顢�Ƶ�����Ϊm1 g���ù����ռ��ˮ���Լ����������ʵ�鷽�����������ʵ�顣
�ͶƲ��ȣ���ѯ��֪п�����ڼZn+2NaOH=Na2ZnO3+H2���ݴˣ���ȡ���ΪS��˫���п���������������顢�Ƶ�����Ϊm1 g���ù����ռ��ˮ���Լ����������ʵ�鷽�����������ʵ�顣
![]() �����ף�ͨ������������Ӧ���ɵ����������ʵ��̽��ľĿ�ꡣ
�����ף�ͨ������������Ӧ���ɵ����������ʵ��̽��ľĿ�ꡣ
![]() ��2��ѡ��B�� ����������ţ�����װ�ý���ʵ�顣
��2��ѡ��B�� ����������ţ�����װ�ý���ʵ�顣
![]() ��3����ó�ַ�Ӧ���������������ΪVL����״������
��3����ó�ַ�Ӧ���������������ΪVL����״������![]() = ��
= ��
![]() ��4������Ʋ��ȣ�����Ҫ������һ���������� ��
��4������Ʋ��ȣ�����Ҫ������һ���������� ��
![]() ��5����װ��B�еĺ�ѹ��Һ©����Ϊ��ͨ��Һ©����������������ƫ����ƫС������Ӱ�족����
��5����װ��B�еĺ�ѹ��Һ©����Ϊ��ͨ��Һ©����������������ƫ����ƫС������Ӱ�족����
![]() �����ң�ͨ������������Ӧǰ�������ʵ��̽��Ŀ�ꡣѡ������C��ʵ�飬��������ַ�Ӧ���˳������ϴ�ӡ���ɣ��Ƶ�������Ϊm2g ��
�����ң�ͨ������������Ӧǰ�������ʵ��̽��Ŀ�ꡣѡ������C��ʵ�飬��������ַ�Ӧ���˳������ϴ�ӡ���ɣ��Ƶ�������Ϊm2g ��
��6��![]() ��
��
��������ͨ������������Ӧǰ���������������Լ��������������ֵ��ΪH2��������ʵ��̽��Ŀ�ꡣʵ��ͬ��ʹ������C��
��7����ʵ�����Ƕȷ����������� �����ң�����ڡ��������ڡ���ͬ�ڡ�����
�鿴�𰸺ͽ���>>
����ѧУ��ѡ - ��ϰ���б� - �����б�
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com