
 8Na2CrO4+2Fe2O3+8CO2
8Na2CrO4+2Fe2O3+8CO2 Na2SO4+Na2Cr2O7+H2O
Na2SO4+Na2Cr2O7+H2O 8Na2CrO4+2Fe2O3+8CO2����Ԫ�ػ��ϼ۵ı仯��������������ԭ��Ӧ����2Na2CrO4+H2SO4
8Na2CrO4+2Fe2O3+8CO2����Ԫ�ػ��ϼ۵ı仯��������������ԭ��Ӧ����2Na2CrO4+H2SO4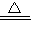 Na2SO4+Na2Cr2O7+H2O��û��Ԫ�ػ��ϼ۵ı仯�����Բ���������ԭ��Ӧ����A����
Na2SO4+Na2Cr2O7+H2O��û��Ԫ�ػ��ϼ۵ı仯�����Բ���������ԭ��Ӧ����A���� 8Na2CrO4+2Fe2O3+8CO2�У���������Ԫ�صõ��ӻ��ϼ۽��ͣ���������������������Ԫ�غ�Ԫ��ʧ���ӻ��ϼ����ߣ����Ի�ԭ����FeO?Cr2O3����B��ȷ��
8Na2CrO4+2Fe2O3+8CO2�У���������Ԫ�صõ��ӻ��ϼ۽��ͣ���������������������Ԫ�غ�Ԫ��ʧ���ӻ��ϼ����ߣ����Ի�ԭ����FeO?Cr2O3����B��ȷ�� 8Na2CrO4+2Fe2O3+8CO2�У�����������������ԭ����FeO?Cr2O3�����������������Դ���Na2CrO4��Fe2O3���������ж�Na2CrO4��Fe2O3�����������ǿ������C����
8Na2CrO4+2Fe2O3+8CO2�У�����������������ԭ����FeO?Cr2O3�����������������Դ���Na2CrO4��Fe2O3���������ж�Na2CrO4��Fe2O3�����������ǿ������C���� 8Na2CrO4+2Fe2O3+8CO2ת�Ƶ��ӿ�֪������1molNa2CrO4ʱ���μӷ�Ӧ��O2�����ʵ���Ϊ
8Na2CrO4+2Fe2O3+8CO2ת�Ƶ��ӿ�֪������1molNa2CrO4ʱ���μӷ�Ӧ��O2�����ʵ���Ϊ mol����Ӧ��OԪ����0�۽���Ϊ-2�ۣ�����ת�Ƶ����ʵ���Ϊ�μӷ�Ӧ�����������ʵ�����4����ת�Ƶ��ӵ����ʵ���Ϊ
mol����Ӧ��OԪ����0�۽���Ϊ-2�ۣ�����ת�Ƶ����ʵ���Ϊ�μӷ�Ӧ�����������ʵ�����4����ת�Ƶ��ӵ����ʵ���Ϊ mol×4=3.5mol�����Է�Ӧ��1����ÿ����1molNa2CrO4ʱ����ת��3.5mol����D����
mol×4=3.5mol�����Է�Ӧ��1����ÿ����1molNa2CrO4ʱ����ת��3.5mol����D����

 ��1����Ԫ�¿�������ĩϵ�д�
��1����Ԫ�¿�������ĩϵ�д�
| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ��Ķ�����
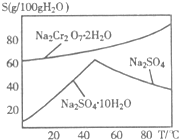 �ظ����ƣ�Na2Cr2O7����Ҫ����ӡȾ���Ƹҽҩ����Ƶȣ���ҵ���Ը�������Ҫ�ɷ�FeO?Cr2O3����̼���ơ�����������Ϊԭ�������ظ����ƣ�Na2Cr2O7?2H2O������Ҫ��Ӧ���£�
�ظ����ƣ�Na2Cr2O7����Ҫ����ӡȾ���Ƹҽҩ����Ƶȣ���ҵ���Ը�������Ҫ�ɷ�FeO?Cr2O3����̼���ơ�����������Ϊԭ�������ظ����ƣ�Na2Cr2O7?2H2O������Ҫ��Ӧ���£�
| ||
. |
| �¶� | 0�� | 100�� |
| �ܽ�� | ||
| ���� | ||
| K2Cr2O7 | 4.6g | 94.1g |
| KCl | 28g | 55g |
| NaCl | 35g | 39g |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
| ||
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
| ���� |
�鿴�𰸺ͽ���>>
����ѧУ��ѡ - ��ϰ���б� - �����б�
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com