
 BaSO4����2H2O��Ba2����SO42��
BaSO4����2H2O��Ba2����SO42�� BaSO4������H����SO42����Ba2����OH��
BaSO4������H����SO42����Ba2����OH�� BaSO4����H2O��H����OH��
BaSO4����H2O��H����OH�� H2O����2Ba2����4OH����Al3+��2 SO42��
H2O����2Ba2����4OH����Al3+��2 SO42�� 2BaSO4����[Al��OH��4]����3[Al��OH��4]����Al3+
2BaSO4����[Al��OH��4]����3[Al��OH��4]����Al3+ 4 Al��OH��3����
4 Al��OH��3���� BaSO4����2H2O����ʱ������Һ��ֻ��Na2SO4��Һ������Ba(OH)2�����ӷ�ӦʽΪBa2����SO42��
BaSO4����2H2O����ʱ������Һ��ֻ��Na2SO4��Һ������Ba(OH)2�����ӷ�ӦʽΪBa2����SO42�� BaSO4�����Ƶ�ǡ�ò������ɳ���Ϊֹʱ�ķ�Ӧ���ӷ���ʽΪH����SO42����Ba2����OH��
BaSO4�����Ƶ�ǡ�ò������ɳ���Ϊֹʱ�ķ�Ӧ���ӷ���ʽΪH����SO42����Ba2����OH�� BaSO4����H2O��ԭ��Һ����NaOH��Һ�������μ�NaHSO4��Һ��ֻ������H����OH��
BaSO4����H2O��ԭ��Һ����NaOH��Һ�������μ�NaHSO4��Һ��ֻ������H����OH�� H2O���ǵ���Һ��Ba2��ǡ����ȫ����ʱ�ķ�Ӧ���ӷ���ʽΪ2Ba2����4OH����Al3+��2 SO42��
H2O���ǵ���Һ��Ba2��ǡ����ȫ����ʱ�ķ�Ӧ���ӷ���ʽΪ2Ba2����4OH����Al3+��2 SO42�� 2BaSO4����[Al��OH��4]����������Һ�м����μ�������Һʱ��3[Al��OH��4]����Al3+
2BaSO4����[Al��OH��4]����������Һ�м����μ�������Һʱ��3[Al��OH��4]����Al3+ 4 Al��OH��3����
4 Al��OH��3����

 �����ҵ���������ϵ�д�
�����ҵ���������ϵ�д� �����̸�Ӯ����ٸ�Ч�����ܸ�ϰ���ϿƼ�������ϵ�д�
�����̸�Ӯ����ٸ�Ч�����ܸ�ϰ���ϿƼ�������ϵ�д� �����ҵ�����������ѧ���ӳ�����ϵ�д�
�����ҵ�����������ѧ���ӳ�����ϵ�д� ����ѧ��Ӯ�����ϵ�д�
����ѧ��Ӯ�����ϵ�д�
| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ������ ���ͣ���ѡ��
A�����ռ���Һ��ͨ������Ķ�����̼��2OH����CO2====CO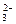 ��H2O ��H2O |
B������Һ��ͨ�������Ķ�����̼��CO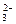 ��CO2��H2O====2HCO ��CO2��H2O====2HCO |
C����ˮ������ͨ�������̼��SiO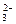 ��CO2��2H2O====H4SiO4����CO ��CO2��2H2O====H4SiO4����CO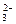 |
D�������������Һ��ͨ�������̼��2ClO����CO2��H2O====2HClO��CO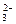 |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ���ѡ��
A����ʯī�缫�����������Һ��4Ag++4OH��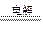 4Ag+ O2��+ 2H2O 4Ag+ O2��+ 2H2O |
| B��̼�������Һ�м������������������Һ��HCO3��+OH����CO32-+ H2O |
| C�����������Һ��ͨ�������Ķ�����̼��Ca2++2C1O��+H2O+CO2��CaCO3��+ 2HClO |
| D������þ��Һ������������Һ��ϣ�Ba2++ SO42����BaSO4�� |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ���ѡ��
A��������С�մ���Һ��Ӧ��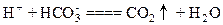 |
| B�����⻯�Ƶ�ˮ�⣺ HS-+H2O 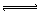 H3O++S2- H3O++S2- |
| C���������Ȼ�����Һ��Ӧ�� Fe+Fe3+====2Fe2+ |
| D����������������Һ��Ӧ�� |
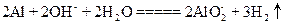
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ���ѡ��
| A���ð�ˮ��������SO2���壺2NH3��H2O+SO2=2NH4++SO32-+H2O |
| B��NaHSO4��Һ�е���Ba(OH)2��Һ�����ԣ�H++SO42-+Ba2+ʮOH-===BaSO4��+H2O |
| C����H2O2�м�������KMnO4��Һ��2MnO4-+5H2O2+6H+====2Mn2+ +5O2��+8H2O |
D���ö��Ե缫��ⱥ���Ȼ�����Һ��2Cl- + 2H+ H2��+ Cl2�� H2��+ Cl2�� |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ���ѡ��

�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ���ѡ��
 CO2��+H2O����ʾ����(����)?
CO2��+H2O����ʾ����(����)? | A��̼�ᱵ�����ᷴӦ? |
| B��̼�����Һ��ϡ���ᷴӦ? |
| C���մ���Һ��ϡ���ᷴӦ? |
| D��С�մ���Һ��ϡ���ᷴӦ? |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ���ѡ��
| A������ͨ�������ҺCH3COOH+NH3=CH3COONH4 |
| B�������ʯ��ˮ�����ᷴӦH++OH-=H2O |
| C��̼�ᱵ���ڴ���BaCO3+2H+=Ba2++H2O+CO2�� |
| D�������Ƹ�ˮ��Ӧ2Na+2H2O=2Na++2OH-+H2�� |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ������
�鿴�𰸺ͽ���>>
����ѧУ��ѡ - ��ϰ���б� - �����б�
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com