
���� ��1�����ݺ�25%NaClO��1000mL���ܶ�1.19g•cm-3��������c=$\frac{1000�Ѧ�}{M}$�����㣻
��2������ϡ��ǰ�����ʵ����ʵ������������㣻
��3��������Һ�����Ƽ�c=$\frac{n}{V}$��m=nM�����㡢������
��� �⣺��1����c��NaClO��=$\frac{1000mL��1.19g•c{m}^{-3}��25%}{74.5g/mol��1L}$=4.0 mol•L-1��
�ʴ�Ϊ��4.0��
��2������ϡ��ǰ�����ʵ����ʵ������䣬��100 mL��4.0 mol•L-1=$\frac{1.19/L��100mL+1g/mL��100mL��100}{1g/mol}$��c��NaClO����
���ϡ�ͺ�c��NaClO����0.04 mol•L-1��c��Na+��=c��NaClO����0.04 mol•L-1��
�ʴ�Ϊ��0.04��
��3��A������������ƽ����NaClO���壬�����ձ����ܽ�NaClO�����ò��������н������������������ƿ�ͽ�ͷ�ι������ݣ�ͼʾ��A��B��C��D����Ҫ�������貣�����ͽ�ͷ�ιܣ���A����
B�����ƹ�������Ҫ����ˮ�����Ծ�ϴ�Ӹɾ�������ƿ���غ�ɺ���ʹ�ã���B����
C������NaClO�����տ����е�H2O��CO2�����ʣ�������ƷNaClO���ܲ��ֱ��ʵ���NaClO���٣����Ƶ���Һ�����ʵ����ʵ�����С�����ƫ�ͣ���C��ȷ��
D��Ӧѡȡ500 mL������ƿ�������ƣ�Ȼ��ȡ��480 mL���ɣ�������ҪNaClO��������0.5 L��4.0 mol•L-1��74.5 g•mol-1=149 g����D����
�ʴ�Ϊ��C��
���� ���⿼���й����ʵ���Ũ�ȵļ��㣬��Ŀ�Ѷ��еȣ���ȷŨ�ȡ���������ʵ����Ĺ�ϵ����Һ���Ƶ�����������ȼ��ɽ�𣬵�����Ϸ�����


 �Ķ��쳵ϵ�д�
�Ķ��쳵ϵ�д�
| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ѡ����
| A�� | CO��HCl | B�� | H2��HCl��CO | C�� | CO2��CO��HCl | D�� | CO2��CO |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������

 ��
���鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
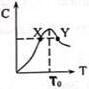 �����ʵ�����Ϊ3.00mol����A��B�����L�����У��������·�Ӧ3A+B?2C���ٷ�Ӧ������C�����ʵ����������¶ȱ仯��ͼ��ʾ��
�����ʵ�����Ϊ3.00mol����A��B�����L�����У��������·�Ӧ3A+B?2C���ٷ�Ӧ������C�����ʵ����������¶ȱ仯��ͼ��ʾ���鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ѡ����
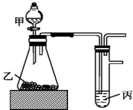 ����ͼװ�ý���ʵ�飬����ʵ���������ý�����ȷ���ǣ�������
����ͼװ�ý���ʵ�飬����ʵ���������ý�����ȷ���ǣ�������| �� | �� | �� | ���� | |
| A | ���� | ʯ��ʯ | Ư����Һ | ���ԣ�HCl��H2CO3��HClO |
| B | ���� | ������ | ����ͭ��Һ | ���ԣ�HCl��H2S��H2SO4 |
| C | ˮ | �������� | ˮ | ����������ˮ�ķ�Ӧ�Ƿ��ȷ�Ӧ |
| D | Ũ���� | �������� | �⻯����Һ | �����ԣ�MnO2��Cl2��I2 |
| A�� | A | B�� | B | C�� | C | D�� | D |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
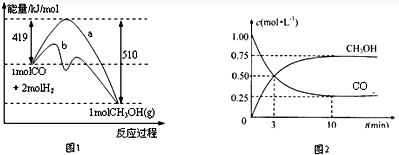
| �¶ȣ�K�� CO2ת���ʣ�%�� n��H2��/n��CO2�� | 500 | 600 | 700 | 800 |
| 1.5 | 45 | 33 | 20 | 12 |
| 2 | 60 | X | 28 | 15 |
| 3 | 83 | 62 | 37 | 22 |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ѡ����
| A�� | ������ϴ�Ӽ������ƹ�ʹ�ã�������Ч����ˮ�帻Ӫ�����ķ��� | |
| B�� | �����Ƿ������绹�ǻ������磬���ǽ���ѧ��ת��Ϊ���� | |
| C�� | PM2.5���е�Ǧ���ӡ�����������ȶ������к���Ԫ�ؾ��ǽ���Ԫ�� | |
| D�� | ���������͵������������γ��������Ҫ���� |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ѡ����
| A�� | A������������C�ֽ��������� | |
| B�� | ��λʱ������nmolA��ͬʱ����3nmolB | |
| C�� | ��λʱ������nmolA��ͬʱ����2nmolC | |
| D�� | ��λʱ������B������������C��������� |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ѡ����
| A�� | �����۷�Ӧ�ų�H2����ɫ��Һ��NO3-��Al3+��Na+��SO42- | |
| B�� | ʹ��ɫʯ����ֽ��������Һ��SO42-��HCO3-��K+��Na+ | |
| C�� | ������Һ��Fe3+��Al3+��NO3-��SO42- | |
| D�� | ������$\frac{c��{H}^{+}��}{c��O{H}^{-}��}$=1��10-12����Һ��K+��AlO2-��CO32-��Na+�� |
�鿴�𰸺ͽ���>>
����ѧУ��ѡ - ��ϰ���б� - �����б�
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com