
���� ��1������Ϊ��©��װҺ�������á���Һ��
��2�����������������²�Һ�壻��G�������ʹҺ��˳�����£�
��3�����ݲ���Ŀ�ķ�����
��4������ˮ�����Ȼ�̼�е��ܽ��Բ�ͬ��
��� �⣺��1������Ϊ��©��װҺ�������á���Һ������ΪC��B��D��A��H��G��E��F��
�ʴ�Ϊ��C��B��D��H��E��F��
��2�����������������²�Һ�壬Ӧʹ©���¶˹ܿڽ����ձ��ڱڣ���ʱ�رջ�������Ҫ���ϲ�Һ����������G������Ŀ��Ϊ����ʱ©����Һ���ܹ�������
�ʴ�Ϊ��ʹ©���¶˹ܿڽ����ձ��ڱڣ���ʱ�رջ�������Ҫ���ϲ�Һ��������
��3����G�����������Ŀ����Ϊ��ʹ©�����������ͨ���Ա�֤����Һ��˳��������
�ʴ�Ϊ��ʹ©�����������ͨ���Ա�֤���У�E������ʱ©����Һ���ܹ�������
��4������ˮ�����ܣ����ҵ��ڱ����ܽ�ȱ���ˮ�д�ö࣬���뱽����Ӧ��
�ʴ�Ϊ���ܣ�����ˮ�����ܣ����ҵ��ڱ����ܽ�ȱ���ˮ�д�ö࣬���뱽����Ӧ��
���� ���⿼���˷�Һ���й�ʵ��������⣬ע��ʵ������IJ��裬�����ѶȲ���



| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ѡ����
| A�� | ����������Һ�м��������ˮ��Al3++3OH-=Al��OH��3�� | |
| B�� | ��Ba��OH��2��Һ�еμ�NaHSO4��Һ�������Һǡ��Ϊ���ԣ�Ba2++OH-+H++SO42-=BaSO4��+H2O | |
| C�� | FeSO4��Һ��ϡ���ᡢ˫��ˮ��ϣ�2Fe2++H2O2+2H+=2Fe3++2H2O | |
| D�� | ��NaHCO3��Һ�м�������Ba��OH��2����Һ��Ba2++2HCO3-+2OH-=2H2O+BaCO3��+CO32- |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ѡ����
| A�� | 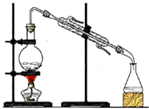 ��ȡ����ˮ | B�� | 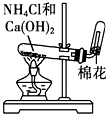 ��ȡNH3 | C�� | 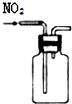 �ռ�NO2 | D�� | 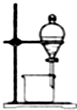 ����ˮ�;ƾ� |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ѡ����
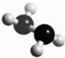 �£�N2H4���ǻ��ȼ�ϣ�����ӵ����ģ����ͼ��ʾ��������H2O2������Ӧ��N2H4+2H2O2�TN2+4H2O����NAΪ����٤��������ֵ������˵����ȷ���ǣ�������
�£�N2H4���ǻ��ȼ�ϣ�����ӵ����ģ����ͼ��ʾ��������H2O2������Ӧ��N2H4+2H2O2�TN2+4H2O����NAΪ����٤��������ֵ������˵����ȷ���ǣ�������| A�� | 32g N2H4�к��й��ۼ�������Ϊ6NA | |
| B�� | ��״���£�22.4LH2O2������ԭ������Ϊ4NA | |
| C�� | 28g N2�к���������Ϊ7NA | |
| D�� | ��17g H2O2�����Ǽ��Լ���Ŀ��ͬ��N2H4�ķ�������Ϊ0.5NA |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ѡ����
| A�� | v��N2����=v��NH3���� | |
| B�� | ��λʱ����3molH-H�����ѣ�ͬʱ6molN-H������ | |
| C�� | n��N2����n��H2������NH3��=1��3��2 | |
| D�� | ���������ܶȲ��ٸı� |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ѡ����
| A�� | �ڹ��ۻ������в����ܺ������Ӽ� | |
| B�� | ��������֮��ͨ�����������γɵĻ�ѧ�����й��ۼ� | |
| C�� | ���й��ۼ��Ļ�����һ���ǹ��ۻ����� | |
| D�� | �������Ӽ��Ļ����ﲻһ�������ӻ����� |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ѡ����
| A�� | �ۢܢݢޢ� | B�� | �ۢܢ� | C�� | �ܢޢ� | D�� | �٢ۢܢ� |
�鿴�𰸺ͽ���>>
����ѧУ��ѡ - ��ϰ���б� - �����б�
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com