
ЁОЬтФПЁПгУЪЏгЭЛЏЙЄВњЦЗ1,3-ЖЁЖўЯЉКЯГЩЛЏЙЄжаМфВњЦЗD ЃЈ![]() ЃЉЕФКЯГЩТЗЯпШчЯТЃК
ЃЉЕФКЯГЩТЗЯпШчЯТЃК
вбжЊЃК

ЃЈ1ЃЉЮяжЪDжаЫљКЌЙйФмЭХУћГЦЮЊ_________ЃЌвЛЖЈЬѕМўЯТЃЌ1 molDзюЖргы_________molH2ЗЂЩњМгГЩЗДгІЁЃ
ЃЈ2ЃЉаДГіЂкЂоЕФЗДгІРраЭЗжБ№ЪЧ_________ЁЂ_________ЁЃ
ЃЈ3ЃЉЂмЗДгІЕФЛЏбЇЗНГЬЪН_________ЁЃЂнЗДгІЕФЛЏбЇЗНГЬЪН_________ЁЃ
ЃЈ4ЃЉA ЕФЯЕЭГУќУћЪЧ_________ЁЃ
ЃЈ5ЃЉаДГігыЮяжЪDЛЅЮЊЭЌЗжвьЙЙЬхЧвТњзувдЯТЬѕМўЕФгаЛњЮяНсЙЙМђЪН_________ЁЃ
Ђй ФмгыFeCl3ШмвКЗЂЩњЯдЩЋЗДгІ
Ђк ФмЗЂЩњвјОЕЗДгІ
Ђл КЫДХЙВеёЧтЦзЯдЪОга5зщЗх
ЃЈ6ЃЉЧыЩшМЦКЯРэЗНАИгУ![]() КЯГЩ
КЯГЩ![]() ЁЃЃЈЦфЫћдСЯздбЁЃЌгУЗДгІСїГЬЭМБэЪОЃЌВЂзЂУїБивЊЕФЗДгІЬѕМўЃЉЁЃ_______
ЁЃЃЈЦфЫћдСЯздбЁЃЌгУЗДгІСїГЬЭМБэЪОЃЌВЂзЂУїБивЊЕФЗДгІЬѕМўЃЉЁЃ_______
ЁОД№АИЁП ЬМЬМЫЋМќ ШЉЛљ 4 МгГЩЗДгІЃЈЛЙдЗДгІЃЉ ЯћШЅЗДгІ CH2(OH)CH2CH2CH2OH+O2 ![]() OHCCH2CH2CHO+2H2O 2OHCCH2CH2CHOЁњ
OHCCH2CH2CHO+2H2O 2OHCCH2CH2CHOЁњ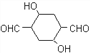 1ЃЌ4-ЖЁЖўДМ
1ЃЌ4-ЖЁЖўДМ 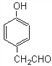
![]()
ЁОНтЮіЁПЗДгІЂлЮЊТБДњЬўЕФЫЎНтЗДгІЩОГ§ДМЃЌAЮЊHOCH2CH2CH2CH2OHЃЌAДпЛЏбѕЛЏЩњГЩШЉЃЌBЮЊOHCCH2CH2CHOЃЌBдкМюадЬѕМўЯТЗЂЩњЗДгІЩњГЩCЃЌCЮЊ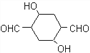 ЃЌИљОнDЕФНсЙЙЃЌCЗЂЩњєЧЛљЕФЯћШЅЗДгІЩњГЩDЁЃ
ЃЌИљОнDЕФНсЙЙЃЌCЗЂЩњєЧЛљЕФЯћШЅЗДгІЩњГЩDЁЃ
(1)D ЮЊ![]() ЃЌЦфжаЫљКЌЙйФмЭХгаЬМЬМЫЋМќКЭШЉЛљЃЌвЛЖЈЬѕМўЯТЃЌШЉЛљКЭЬМЬМЫЋМќОљФмгыЧтЦјМгГЩЃЌ1 molDзюЖргы4molH2ЗЂЩњМгГЩЗДгІЃЌЙЪД№АИЮЊЃКЬМЬМЫЋМќЁЂШЉЛљЃЛ4ЃЛ
ЃЌЦфжаЫљКЌЙйФмЭХгаЬМЬМЫЋМќКЭШЉЛљЃЌвЛЖЈЬѕМўЯТЃЌШЉЛљКЭЬМЬМЫЋМќОљФмгыЧтЦјМгГЩЃЌ1 molDзюЖргы4molH2ЗЂЩњМгГЩЗДгІЃЌЙЪД№АИЮЊЃКЬМЬМЫЋМќЁЂШЉЛљЃЛ4ЃЛ
(2)ЗДгІЂкЮЊЬМЬМЫЋМќЕФМгГЩЗДгІЃЌЗДгІЂоЮЊ ЯћШЅЗДгІЃЌЙЪД№АИЮЊЃКМгГЩЗДгІЃЛЯћШЅЗДгІЃЛ
ЯћШЅЗДгІЃЌЙЪД№АИЮЊЃКМгГЩЗДгІЃЛЯћШЅЗДгІЃЛ
(3)ЗДгІЂмЮЊДМЕФДпЛЏбѕЛЏЃЌЗДгІЕФЛЏбЇЗНГЬЪНЮЊCH2(OH)CH2CH2CH2OH+O2 ![]() OHCCH2CH2CHO+2H2OЁЃЗДгІЂнЕФЛЏбЇЗНГЬЪНЮЊ2OHCCH2CH2CHOЁњ
OHCCH2CH2CHO+2H2OЁЃЗДгІЂнЕФЛЏбЇЗНГЬЪНЮЊ2OHCCH2CH2CHOЁњ ЃЌЙЪД№АИЮЊЃКCH2(OH)CH2CH2CH2OH+O2
ЃЌЙЪД№АИЮЊЃКCH2(OH)CH2CH2CH2OH+O2 ![]() OHCCH2CH2CHO+2H2OЃЛ2OHCCH2CH2CHOЁњ
OHCCH2CH2CHO+2H2OЃЛ2OHCCH2CH2CHOЁњ  ЃЛ
ЃЛ
(4)AЮЊHOCH2CH2CH2CH2OHЃЌЯЕЭГУќУћЮЊ1ЃЌ4-ЖЁЖўДМЃЌЙЪД№АИЮЊЃК1ЃЌ4-ЖЁЖўДМЃЛ
(5)D ЮЊ![]() ЁЃЂй ФмгыFeCl3ШмвКЗЂЩњЯдЩЋЗДгІЃЌЫЕУїКЌгаЗгєЧЛљЃЛЂк ФмЗЂЩњвјОЕЗДгІЃЌЫЕУїКЌгаШЉЛљЃЛЂл КЫДХЙВеёЧтЦзЯдЪОга5зщЗхЃЌТњзуЬѕМўЕФгаЛњЮяЮЊ
ЁЃЂй ФмгыFeCl3ШмвКЗЂЩњЯдЩЋЗДгІЃЌЫЕУїКЌгаЗгєЧЛљЃЛЂк ФмЗЂЩњвјОЕЗДгІЃЌЫЕУїКЌгаШЉЛљЃЛЂл КЫДХЙВеёЧтЦзЯдЪОга5зщЗхЃЌТњзуЬѕМўЕФгаЛњЮяЮЊ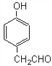 ЃЌЙЪД№АИЮЊЃК
ЃЌЙЪД№АИЮЊЃК ЃЛ
ЃЛ
(6)гУ![]() КЯГЩ
КЯГЩ![]() ЁЃПЩвдЪзЯШНЋ
ЁЃПЩвдЪзЯШНЋ![]() бѕЛЏЮЊєШЫсЃЌдйѕЅЛЏМДПЩЃЌКЯГЩТЗЯпЮЊ
бѕЛЏЮЊєШЫсЃЌдйѕЅЛЏМДПЩЃЌКЯГЩТЗЯпЮЊ![]() ЃЌЙЪД№АИЮЊЃК
ЃЌЙЪД№АИЮЊЃК ЁЃ
ЁЃ



| ФъМЖ | ИпжаПЮГЬ | ФъМЖ | ГѕжаПЮГЬ |
| ИпвЛ | ИпвЛУтЗбПЮГЬЭЦМіЃЁ | ГѕвЛ | ГѕвЛУтЗбПЮГЬЭЦМіЃЁ |
| ИпЖў | ИпЖўУтЗбПЮГЬЭЦМіЃЁ | ГѕЖў | ГѕЖўУтЗбПЮГЬЭЦМіЃЁ |
| ИпШ§ | ИпШ§УтЗбПЮГЬЭЦМіЃЁ | ГѕШ§ | ГѕШ§УтЗбПЮГЬЭЦМіЃЁ |
ПЦФПЃКИпжаЛЏбЇ РДдДЃК ЬтаЭЃК
ЁОЬтФПЁПAЁЂBЁЂCЁЂDЁЂEЪЧЧАЫФжмЦкдзгађЪ§вРДЮдіДѓЕФЮхжждЊЫиЁЃAЁЂDЭЌжїзхЧвФмаЮГЩСНжжГЃМћЛЏКЯЮяDA2КЭDA3ЃЛЛљЬЌCдзгзюЭтЕчзгВуЩЯга1ИіЮДГЩЖдЕчзгЃЛЛљЬЌBЁЂEдзгЕФзюЭтВуОљжЛга2ИіЕчзгЃЌЦфгрИїЕчзгВуОљШЋГфТњЁЃЛиД№ЯТСаЮЪЬтЃК
ЃЈ1ЃЉЛљЬЌDдзгМлЕчзгЕФЙьЕРБэДяЪНЮЊ_________ЃЌдЊЫиЭгыEЕФЕкЖўЕчРыФмЗжБ№ЮЊICu=1985 kJЁЄmol-1ЃЌIE=1733 kJЁЄmol-1ЃЌICu>IEЕФдвђЪЧ_____________________ЁЃ
ЃЈ2ЃЉDA2ЗжзгЕФVSEPRФЃаЭЪЧ_________ЁЃаДГівЛжжгыDA3ЛЅЮЊЕШЕчзгЬхЕФРызгЕФЛЏбЇЪНЃК_____ЁЃ
ЃЈ3ЃЉЪЕбщВтЕУCгыТШдЊЫиаЮГЩЦјЬЌЛЏКЯЮяЕФЪЕМЪзщГЩЮЊC2Cl6ЃЌЦфжаCдзгЕФдгЛЏЗНЪНЮЊ_____________ЁЃвбжЊCCl3дкМгШШЪБвзЩ§ЛЊЃЌгыЙ§СПЕФNaOHШмвКЗДгІПЩЩњГЩ Na[C(OH)4]ЃЌCCl3ЙЬЬхЪєгк______ОЇЬхЃЈЬюОЇЬхРраЭЃЉ
ЃЈ4ЃЉЪдБШНЯAЁЂDМђЕЅЧтЛЏЮяЕФШШЮШЖЈадЃЌВЂЫЕУїРэгЩЃК_________________ЃЈгУМќВЮЪ§НтЪЭЃЉ
ЃЈ5ЃЉDгыEЫљаЮГЩЛЏКЯЮяОЇЬхЕФОЇАћШчЭМЫљЪОЃК

ЂйдкИУОЇЬхжаЃЌDЕФХфЮЛЪ§ЮЊ_____________ЁЃ
ЂкдзгзјБъВЮЪ§ПЩБэЪООЇАћФкВПИїдзгЕФЯрЖдЮЛжУЁЃЩЯЭМОЇАћжаЃЌдзгзјБъВЮЪ§aЮЊ(0ЃЌ0ЃЌ0)ЃЛbЮЊЃЈ![]() ЃЌ0ЃЌ
ЃЌ0ЃЌ ![]() )ЃЛc ЮЊЃЈ
)ЃЛc ЮЊЃЈ![]() ЃЌ
ЃЌ ![]() ЃЌ0)ЁЃдђd ЕФзјБъВЮЪ§ЮЊ_________ЁЃ
ЃЌ0)ЁЃдђd ЕФзјБъВЮЪ§ЮЊ_________ЁЃ
ЂлвбжЊИУОЇЬхЕФУмЖШЮЊІб gЁЄcm-3ЃЌЩшNAБэЪОАЂЗќМгЕТТоГЃЪ§ЕФжЕЃЌдђОЇАћжаDРызггыEРызгжЎМфЕФзюНќОрРыЮЊ__________pmЁЃ
ВщПДД№АИКЭНтЮі>>
ПЦФПЃКИпжаЛЏбЇ РДдДЃК ЬтаЭЃК
ЁОЬтФПЁПвдУКЮЊдСЯПЩКЯГЩвЛЯЕСаШМСЯЁЃ
ЃЈ1ЃЉвбжЊЃКЂй2H2ЃЈgЃЉ+O2ЃЈgЃЉ= 2H2OЃЈgЃЉЁїH=Ѓ483.6kJ/mol
ЂкCH3OH(g)+H2O(g)=CO2(g)+3H2(g)ЁїH=ЃЋ49.0kJ/mol
ЧыаДГіМзДМШМЩеЩњГЩH2OЃЈgЃЉЕФШШЛЏбЇЗНГЬЪН_________;
ЃЈ2ЃЉЯђ1LУмБеШнЦїжаМгШы2mol COЁЂ4mol H2ЃЌдкЪЪЕБЕФДпЛЏМСзїгУЯТЃЌЗЂЩњЗДгІЃК2COЃЈgЃЉ+4H2ЃЈgЃЉ![]() CH3OCH3ЃЈlЃЉ+H2OЃЈlЃЉЁїH=+71kJ/mol
CH3OCH3ЃЈlЃЉ+H2OЃЈlЃЉЁїH=+71kJ/mol
ЂйИУЗДгІФмЗё_________здЗЂНјааЃЈЬюЁАФмЁБЁЂЁАВЛФмЁБЛђЁАЮоЗЈХаЖЯЁБЃЉ
ЂкЯТСаа№ЪіФмЫЕУїДЫЗДгІДяЕНЦНКтзДЬЌЕФЪЧ_________ЃЎ
aЃЎЛьКЯЦјЬхЕФЦНОљЯрЖдЗжзгжЪСПБЃГжВЛБф
bЃЎCOКЭH2ЕФзЊЛЏТЪЯрЕШ
cЃЎCOКЭH2ЕФЬхЛ§ЗжЪ§БЃГжВЛБф
dЃЎЛьКЯЦјЬхЕФУмЖШБЃГжВЛБф
eЃЎ1mol COЩњГЩЕФЭЌЪБга1mol OЃHМќЖЯСб
ЃЈ3ЃЉCO2ЃЈgЃЉ+3H2ЃЈgЃЉ![]() CH3OHЃЈgЃЉ+H2OЃЈgЃЉЁїHЃМ0дквЛЖЈЬѕМўЯТЃЌФГЗДгІЙ§ГЬжаВПЗжЪ§ОнШчЯТБэЃК
CH3OHЃЈgЃЉ+H2OЃЈgЃЉЁїHЃМ0дквЛЖЈЬѕМўЯТЃЌФГЗДгІЙ§ГЬжаВПЗжЪ§ОнШчЯТБэЃК
ЗДгІЬѕМў | ЗДгІЪБМф | CO2(mol) | H2(mol) | CH3OH(mol) | H2O(mol) |
КуЮТ КуШн (T1ЁцЁЂ 2L) | 0min | 2 | 6 | 0 | 0 |
10min | 4.5 | ||||
20min | 1 | ||||
30min | 1 |
Ђй0ЁЋ10minФкЃЌгУH2OЃЈgЃЉБэЪОЕФЛЏбЇЗДгІЫйТЪv(H20)=_________mol/(LЁЄmin)
ЂкДяЕНЦНКтЪБЃЌИУЗДгІЕФЦНКтГЃЪ§K=_________ЃЈгУЗжЪ§БэЪОЃЉЃЌЦНКтЪБH2ЕФзЊЛЏТЪЪЧ_________ЁЃ
ЂлдкЦфЫќЬѕМўВЛБфЕФЧщПіЯТЃЌШє30minЪБИФБфЮТЖШЮЊT
ЃЈ4ЃЉгУМзУбЃЈCH3OCH3ЃЉзїЮЊШМСЯЕчГиЕФдСЯ,ЧыаДГідкМюадНщжЪжаЕчГиИКМЋЗДгІЪН_________ЃЎ
ВщПДД№АИКЭНтЮі>>
ПЦФПЃКИпжаЛЏбЇ РДдДЃК ЬтаЭЃК
ЁОЬтФПЁПФГЭЌбЇЩшМЦСЫВтЖЈЦјЬхФІЖћЬхЛ§ЕФЬНОПЪЕбщЃЌРћгУТШЫсМиЗжНтжЦO2ЁЃ

ЪЕбщВНжшШчЯТЃК
ЂйСЌНгКУЪЕбщзАжУЃЌМьВщзАжУЕФЦјУмадЁЃ
ЂкАбЪЪСПЕФТШЫсМиЗлФЉКЭЩйСПЖўбѕЛЏУЬЗлФЉЛьКЯОљдШЃЌЗХШыИЩдяЕФЪдЙмжаЃЌзМШЗГЦСПЪдЙмКЭвЉЦЗЕФзмжЪСПЮЊ15.95 gЁЃ
ЂлМгШШЃЌПЊЪМЗДгІЃЌжБЕНВЛдйгаЦјЬхВњЩњЮЊжЙЁЃ
ЂмВтСПХХШыСПЭВжаЫЎЕФЬхЛ§ЮЊ285.0 mLЃЌЛЛЫуГЩБъзМзДПіЯТбѕЦјЕФЬхЛ§ЮЊ279. 7 mLЁЃ
ЂнзМШЗГЦСПЪдЙмКЭВаСєЮяЕФжЪСПЮЊ15. 55 gЁЃ
ИљОнЩЯЪіЪЕбщЙ§ГЬЃЌЛиД№ЯТСаЮЪЬтЃК
(1)ШчКЮМьВщзАжУЕФЦјУмадЃП____________________________________________ЁЃ
(2)вдЯТЪЧВтСПЪеМЏЕФЦјЬхЬхЛ§БиаыАќРЈЕФМИИіВНжшЃК
ЂйЕїећСПЭВЕФИпЖШЪЙЙуПкЦПКЭСПЭВФкЕФвКУцИпЖШЯрЭЌЃЛ
ЂкЪЙЪдЙмКЭЙуПкЦПФкЦјЬхЖМРфШДжСЪвЮТЃЛ
ЂлЖСШЁСПЭВФквКЬхЕФЬхЛ§ЁЃ
етШ§ВНВйзїЕФе§ШЗЫГађЪЧ_________________(ЧыЬюаДВНжшДњКХ)ЁЃ
НјааЂлЕФЪЕбщВйзїЪБЃЌШєбіЪгЖСЪ§ЃЌдђЖСШЁбѕЦјЕФЬхЛ§_______(ЬюЁАЦЋДѓЁБЁАЦЋаЁЁБЛђЁАЮогАЯьЁБ)ЁЃ
(3)ЪЕбщЙ§ГЬжаВњЩњбѕЦјЕФЮяжЪЕФСПЪЧ_____molЃЛЪЕбщВтЕУбѕЦјЕФЦјЬхФІЖћЬхЛ§ЪЧ______(БЃСєаЁЪ§ЕуКѓСНЮЛ)ЁЃ
ВщПДД№АИКЭНтЮі>>
ПЦФПЃКИпжаЛЏбЇ РДдДЃК ЬтаЭЃК
ЁОЬтФПЁПТШБНдкШОСЯЁЂвНвЉЙЄвЕжагаЙуЗКЕФгІгУЃЌФГЪЕбщаЁзщРћгУШчЯТзАжУКЯГЩТШБНЃЈжЇГХгУЕФЬњМмЬЈВПЗжЪЁТдЃЉВЂЭЈЙ§вЛЖЈВйзїЬсДПТШБНЁЃ
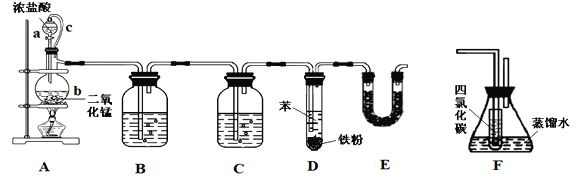
ЗДгІЮяКЭВњЮяЕФЯрЙиЪ§ОнСаБэШчЯТЃК
УмЖШ/gЁЄcmЃ3 | ЗаЕу/Ёц | ЫЎжаШмНтад | |
БН | 0.879 | 80.1 | ЮЂШм |
ТШБН | 1.11 | 131.7 | ВЛШм |
ЧыАДвЊЧѓЛиД№ЯТСаЮЪЬтЁЃ
ЃЈ1ЃЉзАжУAжаЯ№НКЙмcЕФзїгУЪЧ______________ЃЌзАжУEЕФзїгУЪЧ__________________ЁЃ
ЃЈ2ЃЉЪЕбщЪБЃЛЪЙaжаЕФХЈбЮЫсЛКЛКЕЮЯТЃЌПЩЙлВьЕНвЧЦїbФкЕФЯжЯѓЪЧ________________ЃЌаДГіЗДгІЕФРызгЗНГЬЪН______________________________________ЁЃ
ЃЈ3ЃЉЮЊжЄУїТШЦјКЭБНЗЂЩњЕФЪЧШЁДњЖјВЛЪЧМгГЩЗДгІЃЌИУаЁзщгУзАжУFЫЕУїЃЌдђзАжУFжУгк________жЎМфЃЈЬюзжФИЃЉЃЌFжааЁЪдЙмФкCCl4ЕФзїгУЪЧ___________________ЃЌЛЙашЪЙгУЕФЪдМСЪЧ______________ЁЃ
ЃЈ4ЃЉвбжЊDжаМгШы5 mLБНЃЌОЙ§ЬсДПКѓЪеМЏЕНТШБН3.0 gЃЌдђТШБНЕФВњТЪЮЊ_________%ЃЈБЃСєШ§ЮЛгааЇЪ§зжЃЉЁЃ
ВщПДД№АИКЭНтЮі>>
ПЦФПЃКИпжаЛЏбЇ РДдДЃК ЬтаЭЃК
ЁОЬтФПЁПЃЈ1ЃЉЧыИљОнЙйФмЭХЕФВЛЭЌЖдЯТСагаЛњЮяНјааЗжРрЃК
Ђй(CH3)3CCH2OH Ђк Ђл
Ђл![]()
Ђм![]() Ђн
Ђн![]() Ђо
Ђо![]()
ЩЯЪіЮяжЪжаЃЌЪєгкЗМЯуДМЕФЪЧ_________ЃЌЪєгкЗгРрЕФЪЧ___________
ЪєгкєШЫсРрЕФЪЧ__________ЃЌЪєгкШЉРрЕФЪЧ__________ЬюађКХ
ЃЈ2ЃЉЯТСаЮяжЪжаЛЅЮЊЭЌЗжвьЙЙЬхЕФга__________ЃЌЛЅЮЊЭЌЫивьаЮЬхЕФга______ЃЌ
ЛЅЮЊЭЌЮЛЫиЕФга_____________ЃЌЪЧЭЌвЛжжЮяжЪЕФга________(ЬюађКХ)ЁЃ

(3)АДвЊЧѓаДГіЯТСаЛЏбЇЗНГЬЪНЁЃ
Ђй МзБНЁњTNT ___________________________ЃЛ
Ђк 2-фхБћЭщЁњБћЯЉ____________________________ЃЛ
ВщПДД№АИКЭНтЮі>>
ПЦФПЃКИпжаЛЏбЇ РДдДЃК ЬтаЭЃК
ЁОЬтФПЁПЖЬжмЦкдЊЫиRЁЂTЁЂQЁЂWдкдЊЫижмЦкБэжаЕФЯрЖдЮЛжУШчгвЯТЭМЫљЪОЃЌЦфжаTЫљДІЕФжмЦкађЪ§гызхађЪ§ЯрЕШЁЃЯТСаХаЖЯВЛе§ШЗЕФЪЧ

A. зюМђЕЅЦјЬЌЧтЛЏЮяЕФШШЮШЖЈадЃКR>Q
B. зюИпМлбѕЛЏЮяЖдгІЫЎЛЏЮяЕФЫсадЃКQ<W
C. дзгАыОЖЃКT>Q>R
D. КЌTЕФбЮШмвКвЛЖЈЯдЫсад
ВщПДД№АИКЭНтЮі>>
ПЦФПЃКИпжаЛЏбЇ РДдДЃК ЬтаЭЃК
ЁОЬтФПЁПGaNЁЂGaPЁЂGaAsЪЧШЫЙЄКЯГЩЕФвЛЯЕСааТаЭАыЕМЬхВФСЯЃЌЦфОЇЬхНсЙЙОљгыН№ИеЪЏЯрЫЦЁЃЭЪЧживЊЕФЙ§ЖЩдЊЫиЃЌФмаЮГЩЖржжХфКЯЮяЃЌШчCu2ЃЋгыввЖўАЗЃЈH2N-CH2-CH2-NH2ЃЉПЩаЮГЩШчЭМЫљЪОХфРызгЁЃЛиД№ЯТСаЮЪЬтЃК

ЃЈ1ЃЉЛљЬЌGaдзгМлЕчзгЕФЙьЕРБэДяЪНЮЊ________________ЃЛ
ЃЈ2ЃЉШлЕуЃКGaN_____GaPЃЈЬюЁА>ЁБЛђЁА<ЁБЃЉЃЛ
ЃЈ3ЃЉЕквЛЕчРыФмЃКAs_____SeЃЈЬюЁА>ЁБЛђЁА<ЁБЃЉЃЛ
ЃЈ4ЃЉCu2ЃЋгыввЖўАЗЫљаЮГЩЕФХфРызгФкВПВЛКЌгаЕФЛЏбЇМќРраЭЪЧ______ЃЛ
aЃЎХфЮЛМќ b.МЋадМќ cЃЎРызгМќ dЃЎЗЧМЋадМќ
ЃЈ5ЃЉввЖўАЗЗжзгжаЕЊдзгЙьЕРЕФдгЛЏРраЭЮЊ________ЃЌввЖўАЗКЭШ§МзАЗ[N(CH3)3]ОљЪєгкАЗЁЃЕЋввЖўАЗБШШ§МзАЗЕФЗаЕуИпКмЖрЃЌдвђЪЧ___________ЃЛ
ЃЈ6ЃЉCuЕФФГжжОЇЬхОЇАћЮЊУцаФСЂЗННсЙЙЃЌОЇАћБпГЄЮЊacmЃЌЭдзгЕФАыОЖЮЊrcmЁЃИУОЇЬхжаЭдзгЕФЖбЛ§ЗНЪНЮЊ_______аЭЃЈЬюЁАA1ЁБЁЂЁАA2ЁБЛђЁАA3ЁБЃЉЃЌИУОЇЬхУмЖШЮЊ____g/cm3ЃЈгУКЌaКЭNAЕФДњЪ§ЪНБэДяЃЉЃЌИУОЇЬхжаЭдзгЕФПеМфРћгУТЪЮЊ______ЃЈгУКЌaКЭrЕФДњЪ§ЪНБэДяЃЉЁЃ
ВщПДД№АИКЭНтЮі>>
ПЦФПЃКИпжаЛЏбЇ РДдДЃК ЬтаЭЃК
ЁОЬтФПЁПдкАБЫЎжаЃЌNH3ЁЄH2OЕФЕчРыДяЕНЦНКтЕФБъжОЪЧЃЈ ЃЉ
A. ШмвКЯдЕчжаадB. ШмвКжаЮоNH3ЁЄH2OЗжзг
C. ЧтбѕИљРызгХЈЖШКуЖЈВЛБфD. ШмвКжаNH3ЁЄH2OгыNH4+КЭOH-ЙВДц
ВщПДД№АИКЭНтЮі>>
ЙњМЪбЇаЃгХбЁ - СЗЯАВсСаБэ - ЪдЬтСаБэ
КўББЪЁЛЅСЊЭјЮЅЗЈКЭВЛСМаХЯЂОйБЈЦНЬЈ | ЭјЩЯгаКІаХЯЂОйБЈзЈЧј | ЕчаХеЉЦОйБЈзЈЧј | ЩцРњЪЗащЮожївхгаКІаХЯЂОйБЈзЈЧј | ЩцЦѓЧжШЈОйБЈзЈЧј
ЮЅЗЈКЭВЛСМаХЯЂОйБЈЕчЛАЃК027-86699610 ОйБЈгЪЯфЃК58377363@163.com