
(I) ���ӹ�ҵ����30����FeCl3��Һ��ʴ���ھ�Ե���ϵ�ͭ��������ӡˢ��·�塣��FeCl3��Һ����ʴҺ��Cu��Ӧ����CuCl2��FeCl2��
(1) д���÷�Ӧ�Ļ�ѧ����ʽ ��
(2) ������Һ��Fe3+���ڵ��Լ��� ��֤��Fe3+���ڵ������� ��
(��) ӡˢ��·�ķϸ�ʴҺ���д���CuCl2��FeCl2��FeCl3�������ŷŻ���ɻ�����Ⱦ����Դ���˷ѡ�ͨ���������̿ɴӸ÷�Һ�л���ͭ���������Ļ�����ȫ��ת��ΪFeCl3��Һ����Ϊ��ʴҺԭ��ѭ��ʹ�á�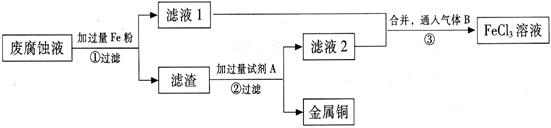
�� ������з�����Ӧ�����ӷ���ʽ ��
(2) ������������Լ�A�� (�ѧʽ)��
(3) �����ͨ�������B�� (�ѧʽ)��д���÷�Ӧ�Ļ�ѧ����ʽ ��
(4) Ϊ�ⶨ������ͭ������������ȡ50g���������������Լ�A��Ӧ���õ�5.6L���壨��״���£����Լ���������ͭ������������
��16�֣�
(I) (1)2FeCl3+Cu==2FeCl2��CuCl2 (2��) [��ѧʽ��������]
(2)KSCN��Һ(1��)�� ��Һ���Ѫ��ɫ(1��)
(II) (1)2Fe3++Fe��3Fe2+ (2��) Fe+Cu2+��Fe2++Cu (2��) [��ѧʽ��������]
(2) HCl (1��)
(3) C12 (1��) 2FeCl2+C12��2FeCl3 (2��) [��ѧʽ��������]
(4) 72% ��4�֣��������ⲽ����֣�
����ʽռ1��
������������������ʵ���ռ1�֣�ռ2��
���ͭ����������ռ1��
�������������(I) (1)���������ṩ�ķ�Ӧ���������ʷ�Ӧ�Ļ�ѧ����ʽΪ2FeCl3+Cu==2FeCl2��CuCl2��
(2)����Fe3+������������Ӧ�����Լ���KSCN��Һ����������Һ���Ѫ��ɫ��
(��) ��Fe�ۻ��CuCl2��FeCl3��Ӧ�������ӷ���ʽΪ2Fe3++Fe��3Fe2+ Fe+Cu2+��Fe2++Cu��
(2)������Fe���������У�����HClʹ�䷴Ӧ���ʼ�����Լ�A��HCl��
(3)��Һ1����Һ2�ж���Fe2+���ӣ���������ʹ�䷴Ӧ����Fe3+����ͨ�������B��C12����Ӧ�Ļ�ѧ����ʽΪ2FeCl2+C12��2FeCl3��
(4)���ݷ�Ӧ����ʽFe+Cu2+��Fe2++Cu Fe+2HCl= FeCl2+H2�����㡣
���㣺�������Ļ����֮ͭ��ķ�Ӧ
�������������й�ʵ�鷽������ƺ�����Ӧ�Ŀ��飬Ҫ��ѧ����Ϥ��ʵ������ݼ�ԭ�����ܹ�����ͬѧ�ǽ��з������⡢��������������



| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ��2011-2012ѧ�����Ͼ����һ��ѧ��һ����ĩ���Ի�ѧ�Ծ����������� ���ͣ�ʵ����
(I)���ӹ�ҵ����30����FeCl3����Һ��ʴ���ھ�Ե���ϵ�ͭ��������ӡˢ��·�塣��FeCl3��Һ����ʴҺ��Cu��Ӧ����CuCl2��FeCl2��
(1)д���÷�Ӧ�Ļ�ѧ����ʽ ��
(2)������Һ��Fe3+���ڵ��Լ���
(��)ӡˢ��·�ķϸ�ʴҺ���д���CuCl2��FeCl2��FeCl3�������ŷŻ���ɻ�����Ⱦ����Դ���˷ѡ�ͨ���������̿ɴӸ÷�Һ�л���ͭ���������Ļ�����ȫ��ת��ΪFeCl3��Һ����Ϊ��ʴҺԭ��ѭ��ʹ�á�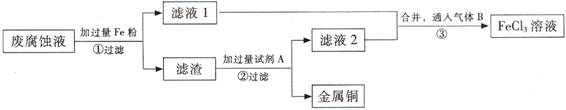 (3) ������з�����Ӧ�����ӷ���ʽ
(3) ������з�����Ӧ�����ӷ���ʽ
(4)��������Ҫ�ɷ��� �� (�ѧʽ)��
(5)Ϊ�˳�ȥ�����е����ʵõ�ͭ���������Լ�A�� (�ѧʽ)��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2015��㶫½����ʯ��ѧ��һ��ѧ�ڵ�һ���¿���ѧ�Ծ��������棩 ���ͣ�ʵ����
(I) ���ӹ�ҵ����30����FeCl3��Һ��ʴ���ھ�Ե���ϵ�ͭ��������ӡˢ��·�塣��FeCl3��Һ����ʴҺ��Cu��Ӧ����CuCl2��FeCl2��
(1) д���÷�Ӧ�Ļ�ѧ����ʽ ��
(2) ������Һ��Fe3+���ڵ��Լ��� ��֤��Fe3+���ڵ������� ��
(��) ӡˢ��·�ķϸ�ʴҺ���д���CuCl2��FeCl2��FeCl3�������ŷŻ���ɻ�����Ⱦ����Դ���˷ѡ�ͨ���������̿ɴӸ÷�Һ�л���ͭ���������Ļ�����ȫ��ת��ΪFeCl3��Һ����Ϊ��ʴҺԭ��ѭ��ʹ�á�

�� ������з�����Ӧ�����ӷ���ʽ ��
(2) ������������Լ�A�� (�ѧʽ)��
(3) �����ͨ�������B�� (�ѧʽ)��д���÷�Ӧ�Ļ�ѧ����ʽ ��
(4) Ϊ�ⶨ������ͭ������������ȡ50g���������������Լ�A��Ӧ���õ�5.6L���壨��״���£����Լ���������ͭ������������
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2014�����Ͼ����һ��ѧ��һ����ĩ���Ի�ѧ�Ծ��������棩 ���ͣ�ʵ����
(I)���ӹ�ҵ����30����FeCl3����Һ��ʴ���ھ�Ե���ϵ�ͭ��������ӡˢ��·�塣��FeCl3��Һ����ʴҺ��Cu��Ӧ����CuCl2��FeCl2��
(1)д���÷�Ӧ�Ļ�ѧ����ʽ ��
(2)������Һ��Fe3+���ڵ��Լ���
(��)ӡˢ��·�ķϸ�ʴҺ���д���CuCl2��FeCl2��FeCl3�������ŷŻ���ɻ�����Ⱦ����Դ���˷ѡ�ͨ���������̿ɴӸ÷�Һ�л���ͭ���������Ļ�����ȫ��ת��ΪFeCl3��Һ����Ϊ��ʴҺԭ��ѭ��ʹ�á�
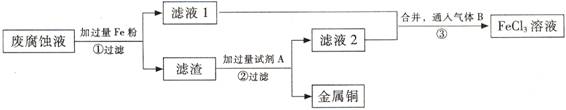 (3) ������з�����Ӧ�����ӷ���ʽ
(3) ������з�����Ӧ�����ӷ���ʽ
(4)��������Ҫ�ɷ��� �� (�ѧʽ)��
(5)Ϊ�˳�ȥ�����е����ʵõ�ͭ���������Լ�A�� (�ѧʽ)��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
(I)���ӹ�ҵ����30����FeCl3����Һ��ʴ���ھ�Ե���ϵ�ͭ��������ӡˢ��·�塣��FeCl3��Һ����ʴҺ��Cu��Ӧ����CuCl2��FeCl2��
(1)д���÷�Ӧ�Ļ�ѧ����ʽ ��
(2)������Һ��Fe3+���ڵ��Լ���
֤��Fe2+���ڵ������� ��
(��)ӡˢ��·�ķϸ�ʴҺ���д���CuCl2��FeCl2��FeCl3�������ŷŻ���ɻ�����Ⱦ����Դ���˷ѡ�ͨ���������̿ɴӸ÷�Һ�л���ͭ���������Ļ�����ȫ��ת��ΪFeCl3 ��Һ����Ϊ��ʴҺԭ��ѭ��ʹ�á�

(3) ������з�����Ӧ�����ӷ�
(4)��������Ҫ�ɷ��� (�ѧʽ)��
(5)Ϊ�˳�ȥ�����е����ʵõ�ͭ���������Լ�A�� (�ѧʽ)��
(6)�����ͨ�˵�����B (�ѧʽ)��
д���÷�Ӧ�Ļ�ѧ����ʽ ��
�鿴�𰸺ͽ���>>
����ѧУ��ѡ - ��ϰ���б� - �����б�
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com