
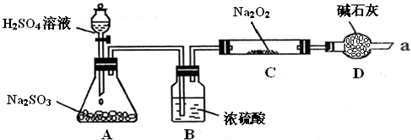
��14�֣�ijͬѧ��ͨ��������ͼ��ʾװ�ã��г�װ����ȥ��ʵ�飬̽��SO2��Na2O2��Ӧ�IJ���

���������װ��B������ װ��D������
����μ��鷴Ӧ���Ƿ���O2����
��C�й������������¼��裺����1��ֻ��Na2SO3 ����2��ֻ��Na2SO4
����3��
��1����������2���У���Ӧ����ʽΪ
��2����Na2O2��Ӧ��ȫ��Ϊȷ��C�й������ijɷ֣���ͬѧ�������ʵ�飺
�ó����ۣ�����2����
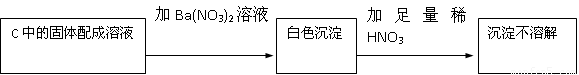
�÷����Ƿ���� ����ǡ�����������
��3��������2��������Ӧǰ��C������6.4g �������10g�������ʵ�Na2O2�����У�Na2O2����������Ϊ �����������ʲ���SO2������Ӧ��
 ��������ѧ����ϵ�д�
��������ѧ����ϵ�д� ��Ԫ������ĩ��ϵ�д�
��Ԫ������ĩ��ϵ�д�
| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ��Ķ�����

| m2-m1 |
| 64 |
| m2-m1 |
| 64 |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
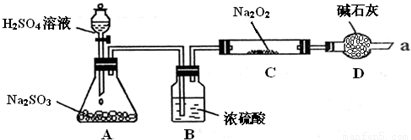
��14�֣�ijͬѧ��ͨ��������ͼ��ʾװ�ã��г�װ����ȥ��ʵ�飬̽��SO2��Na2O2��Ӧ�IJ���
���������װ��B������ װ��D������
����μ��鷴Ӧ���Ƿ���O2����
��C�й������������¼��裺����1��ֻ��Na2SO3 ����2��ֻ��Na2SO4
����3��
��1����������2���У���Ӧ����ʽΪ
��2����Na2O2��Ӧ��ȫ��Ϊȷ��C�й������ijɷ֣���ͬѧ�������ʵ�飺
�ó����ۣ�����2����
�÷����Ƿ���� ����ǡ�����������
��3��������2��������Ӧǰ��C������6.4g �������10g�������ʵ�Na2O2�����У�Na2O2����������Ϊ �����������ʲ���SO2������Ӧ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2011-2012ѧ��㶫ʡ��ͷ�н�ɽ��ѧ��һ12���¿���ѧ�Ծ� ���ͣ�ʵ����
��14�֣�ijͬѧ��ͨ��������ͼ��ʾװ�ã��г�װ����ȥ��ʵ�飬̽��SO2��Na2O2��Ӧ�IJ���
���������װ��B������ װ��D������
����μ��鷴Ӧ���Ƿ���O2����
��C�й������������¼��裺����1��ֻ��Na2SO3 ����2��ֻ��Na2SO4
����3��
��1����������2���У���Ӧ����ʽΪ
��2����Na2O2��Ӧ��ȫ��Ϊȷ��C�й������ijɷ֣���ͬѧ�������ʵ�飺
�ó����ۣ�����2����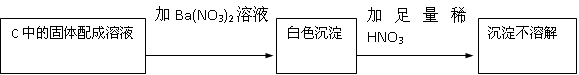
�÷����Ƿ���� ����ǡ�����������
��3��������2��������Ӧǰ��C������6.4g �������10g�������ʵ�Na2O2�����У�Na2O2����������Ϊ �����������ʲ���SO2������Ӧ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2010�긣��ʡ����ʵ����ѧ�߿���ѧģ�����һ���������棩 ���ͣ������

�鿴�𰸺ͽ���>>
����ѧУ��ѡ - ��ϰ���б� - �����б�
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com