
 ��ķ���ʽΪ ��
��ķ���ʽΪ �� ��
��
| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ������ ���ͣ���ѡ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ���ѡ��
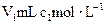 �İ�ˮ�μӵ�
�İ�ˮ�μӵ�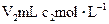 �������У�������������ȷ���ǣ�����
�������У�������������ȷ���ǣ�����A���������Һ��pH��7����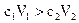 |
B���� �� �� ������Һ�� ������Һ��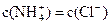 |
C�������Һ��pH��7������Һ��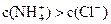 |
D���� ���һ��Һ��pH��7����һ���� ���һ��Һ��pH��7����һ����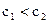 |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ������
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ������
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ���ѡ��
A����a=b���� ������ͬ����ʱ������ ������ͬ����ʱ������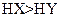 |
B����a=b���� ������ͬ����ʱ������ ������ͬ����ʱ������ |
C����a>b���� ������ͬ����ʱ������HX<HY ������ͬ����ʱ������HX<HY |
D�����������ϣ���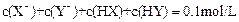 ����һ���� ����һ���� |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ�������
 ��Ũ��Һ������Ӧ��ȫ���ϲ���Һ��Ba2+Ũ�ȱ�Ϊ����?[����仯���Բ��ƣ�Ksp(BaSO4)=1��08��10-10]
��Ũ��Һ������Ӧ��ȫ���ϲ���Һ��Ba2+Ũ�ȱ�Ϊ����?[����仯���Բ��ƣ�Ksp(BaSO4)=1��08��10-10]�鿴�𰸺ͽ���>>
����ѧУ��ѡ - ��ϰ���б� - �����б�
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com