
(12��)������һ���¶��µ�ijNaOH��Һ����֪:����Һ���ΪV mL����Һ�ܶ�Ϊd g/cm3����������(�����ٷֱ�Ũ��)Ϊw%�����ʵ���Ũ��Ϊc mol/L����Һ�к��������Ƶ�����Ϊm g. �Իش��������⣮
(1)���㣺��w��d��ʾ��Һ�����ʵ����ʵ���Ũ��cΪ________________��
(2)ijѧ����������ƽ����С�ձ�������(�ձ���ʢNaOH)������ǰ��������ڱ�ߵ���̶ȣ���ƽ��ֹʱ����ָ���ڷֶ��̵�ƫ��λ�ã���ʱ��ߵ����̽�________________(����ڡ����ڡ�)�ұߵ����̣���ʹ��ƽƽ�⣬�����еIJ���Ϊ____________ ________________ ���ٶ����ճ���С�ձ�������Ϊ________________(�32.6 g����32.61 g��)��
(3)�ڱ��(ͼ)�ϻ�������λ��(����������ʾ)��

(4)���ݺ���Һ���ȣ�����ʱ����Һ����ڿ̶��ߣ������ּ�����ˮ���̶��ߣ�������Һ�����ʵ���Ũ�Ƚ�__________________(�ƫ����ƫС������Ӱ�족)��
(1) c��0.25dw mol/L��(2)���� ����������ҩƷ. 32.6 g
(3) 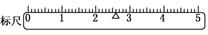 ��4��ƫС (ÿ��2��)
��4��ƫС (ÿ��2��)
��������(1)����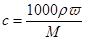 ��֪��
��֪��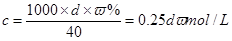 ��
��
��2��ָ���ڷֶ��̵�ƫ��λ�ã�˵���Ҷ�����������˵ģ������ߵ����̽������ұߵ����̣���ʹ��ƽƽ�⣬�����еIJ���Ӧ��������������ҩƷ��������ƽֻ�ܶ�����0.1g�����Դ�ѡ32.6 g��
��3�����������֪�������������2.6g�����Դ��� ��
��
��4���ּ�����ˮ���̶��ߣ�˵����Һ�����ƫ��Ũ��ƫС��



| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ��2011-2012����ױ���������ʯ��ҵһ�и߶���ѧ����ĩ���Ի�ѧ�Ծ����������� ���ͣ�ʵ����
(12��)������һ���¶��µ�ijNaOH��Һ����֪:����Һ���ΪV mL����Һ�ܶ�Ϊd g/cm3����������(�����ٷֱ�Ũ��)Ϊw%�����ʵ���Ũ��Ϊc mol/L����Һ�к��������Ƶ�����Ϊm g. �Իش��������⣮
(1)���㣺��w��d��ʾ��Һ�����ʵ����ʵ���Ũ��cΪ________________��
(2)ijѧ����������ƽ����С�ձ�������(�ձ���ʢNaOH)������ǰ��������ڱ�ߵ���̶ȣ���ƽ��ֹʱ����ָ���ڷֶ��̵�ƫ��λ�ã���ʱ��ߵ����̽�________________(����ڡ����ڡ�)�ұߵ����̣���ʹ��ƽƽ�⣬�����еIJ���Ϊ____________ ________________ ���ٶ����ճ���С�ձ�������Ϊ________________(�32.6 g����32.61 g��)��
(3)�ڱ��(ͼ)�ϻ�������λ��(����������ʾ)��
(4)���ݺ���Һ���ȣ�����ʱ����Һ����ڿ̶��ߣ������ּ�����ˮ���̶��ߣ�������Һ�����ʵ���Ũ�Ƚ�__________________(�ƫ����ƫС������Ӱ�족)��
�鿴�𰸺ͽ���>>
����ѧУ��ѡ - ��ϰ���б� - �����б�
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com