��CO��H
2���ϳɼ״��������������õķ���֮һ��
��1��������Ӧ�Ĵ������õ����������ֽ���Ԫ�صĻ��������һ��Ԫ�ص�ԭ��L�������������������֮��Ϊ4��1��d����еĵ�����������������֮��Ϊ5��1������Ԫ�ط���Ϊ______����+2�����ӵĺ�������Ų�ʽ��______��
��2�����ݵȵ���ԭ����д��CO���ӵĽṹʽ______��
��3���״��������ɵõ���ȩ��
�ټ״��ķе�ȼ�ȩ�ĸߣ�����Ҫԭ����______��
�ڼ�ȩ������̼ԭ�ӹ�����ӻ�����Ϊ______����ȩ���ӵĿռ乹����______�� 1mol��ȩ�����ЦҼ�����ĿΪ______��
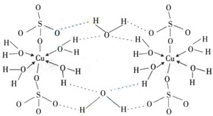
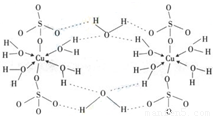
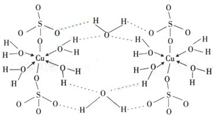
��4��������Ӧ�Ĵ�����һ����ͭԪ�صĻ������֪ͭ����Ҫ�����ﵨ��CuSO
4?5H
2O���[Cu��H
2O��
4]SO
4?H
2O����ṹʾ��ͼ�磺����˵����ȷ����______������ĸ����
A���������ṹʾ��ͼ�У�������ԭ�Ӷ�����sp
3�ӻ�
B���������ṹʾ��ͼ�У�������λ�������ۼ������Ӽ�
C�������Ƿ��Ӿ��壬���Ӽ�������
D�������е�ˮ�ڲ�ͬ�¶��»�ֲ�ʧȥ��

 ��CO��H2���ϳɼ״��������������õķ���֮һ��
��CO��H2���ϳɼ״��������������õķ���֮һ��

 �Ͻ�ƽ��У����ϵ�д�
�Ͻ�ƽ��У����ϵ�д�
 ��2010?����ģ�⣩��CO��H2���ϳɼ״��������������õķ���֮һ��
��2010?����ģ�⣩��CO��H2���ϳɼ״��������������õķ���֮һ��