
 �Ķ��쳵ϵ�д�
�Ķ��쳵ϵ�д�
| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ��Ķ�����
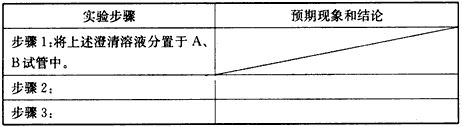
 ��2012?����һģ��ij�о���ѧϰС�����о���������Ư������ʱ���ӡ�������Ư������ʵ������������ˮ��Ӧ���ɵĴ������Ư�����á��õ�������Ϊ��̽�������������õõ����Ƕ������������Ƕ���������ˮ���õIJ����С�����������ʵ�飮��ش�������⣮
��2012?����һģ��ij�о���ѧϰС�����о���������Ư������ʱ���ӡ�������Ư������ʵ������������ˮ��Ӧ���ɵĴ������Ư�����á��õ�������Ϊ��̽�������������õõ����Ƕ������������Ƕ���������ˮ���õIJ����С�����������ʵ�飮��ش�������⣮�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ��Ķ�����

�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
 ij�о���ѧϰС�����о���������Ư������ʱ���ӡ�������Ư������ʵ������������ˮ��Ӧ���ɵĴ������Ư�����á��õ�������Ϊ��̽�������������õõ����Ƕ������������Ƕ���������ˮ���õIJ����С�����������ʵ�飮��ش�������⣮
ij�о���ѧϰС�����о���������Ư������ʱ���ӡ�������Ư������ʵ������������ˮ��Ӧ���ɵĴ������Ư�����á��õ�������Ϊ��̽�������������õõ����Ƕ������������Ƕ���������ˮ���õIJ����С�����������ʵ�飮��ش�������⣮�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������һģ ���ͣ��ʴ���
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ��Ķ�����
ijѧϰС�����о�CH4��ԭFe2O3��ʵ���У��������ɵĺ�ɫ��ĩ����־��ܱ������������������ϵ�֪�����¶Ȳ�ͬ�����Ȳ���ʱ�����ɾ��д��Ե�Fe3O4������൱��Fe2O3•FeO����Ϊ��һ��̽����ɫ��ĩ����ɼ����������ǽ���������ʵ�顣
һ�����Լ���
��1������ɫ��ĩ�еμ����ᣬ���������ݲ��������ɫ��ĩ��һ����_ ___���������ݵ����ӷ���ʽΪ____ ��
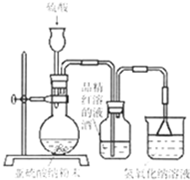
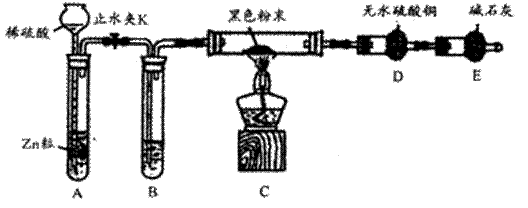
��2���Ȼ�ԭ�����飺����ͼװ�����Ӻ�������ͼ�мг��豸����ȥ�������װ�õ������ԡ���װ��������ҩƷ����ֹˮ��Kͨ������������һ��ʱ����ٵ�ȼC���ľƾ��ơ�
��ش��������⣺
�ٵ�ȼC���ƾ���֮ǰ��Ϊ��Ҫ�ȳ���ͨH2һ��ʱ�� ��
��B���Լ���������_ ��
��������һ��ʱ���װ��D�а�ɫ��ĩ����ɫ���Ժ�ɫ��ĩ����ɵó��Ľ����� ��
��3��Ϊ֤����ɫ��ĩ�Ƿ�Fe3O4��ijͬѧ������������ʵ�飺ȡ������ɫ��ĩ���Թܣ����������ᣬ�����ʹ��ɫ��ĩ��ȫ�ܽ⣬������Һ�����ݣ���һ����Һ����KSCN��Һ���۲���Һ�Ƿ��죻�ڶ�����Һ��������KMnO4��Һ���۲�����KMnO4��Һ�Ƿ���ɫ����ش𣺵�һ����Һ����KSCN��Һ��һ���������������ӷ���ʽ��� ���ڶ�����Һ�����еIJ�������������Һ���Ƿ���� ���ӡ�������������Ϊ�˷����Ƿ���� ����ǡ�����
���������������ⶨ
������ʵ�����̽��вⶨ��
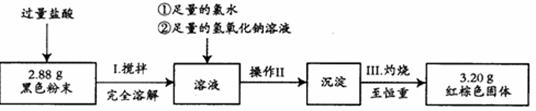
��4������II��������________ �� ____ ��ת�ƹ��塣
��5��ͨ���������ݣ������ɫ��ĩ�и���ֵ����ʵ���֮��Ϊ ��
�鿴�𰸺ͽ���>>
����ѧУ��ѡ - ��ϰ���б� - �����б�
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com