
��һ�� ������500mL0.2mol/LNa2CO3��Һ���ش��������⣺
��1��Ӧ��ȡ g Na2CO3��10H2O��
��2�����Ƹ���Һ���õ������� ____ ��
��3�����в���ʹ������ҺŨ��ƫ�͵��ǣ� ��
A������ƿδ���T����������Һ B��������ƿת����Һʱ������Һ�彦��
C��δϴ���ܽ������ձ� D������ʱ�����ӿ̶���
������ij��ѧС����ʵ��������ͼ�ṩ�������Ʊ������顣
��֪��
�ٷ�Ӧԭ����NaBr+H2SO4����Ũ����=== NaHSO4+HBr
HBr+C2H5OHC2H5Br+H2O
�ڷ�Ӧ��������NaBr(S)25g����ˮ�Ҵ�15mL��ŨH2SO4 30mL��ˮ15mL
����������Ҵ��IJ��������������±�
|
| �ܶ�/g��mL-1 | �е�/�� | �ܽ��� |
| ������ | 1.461 | 38 | ������ˮ |
| �Ҵ� | 0.789 | 78 | ������ˮ |
�ش��������⣺
��4����������������˳���ǣ�1��( )��( )��(4 )��( 3 )��( )�������֣�������Ϊ��Ӧ���������� �����ƿ�����ձ�������
��5������Ũ�������ǿ�����ԣ���Ӧ���������ʹ���������鳣�ʻ�ɫ���ø������� ��д��ѧʽ������ȥ�����ʵ��Լ��ͷ����� ��Ϊ�˼��ٸø���������ɣ����ݼ���ķ�Ӧ���ʵ���ȡ�� _________________________________________________________�Ĵ�ʩ��

| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ��2016���Ĵ�ʡ��һ��ѧ�����п��Ի�ѧ�Ծ��������棩 ���ͣ������
��ͼΪʵ����ijŨ�����Լ�ƿ��ǩ�ϵ��й����ݣ��Ը��ݱ�ǩ�ϵ��й����ݻش��������⣺

��1����Ũ������HCl�����ʵ���Ũ��Ϊ__________mol��L-1��
��2��ȡ����������ĸ�������Һʱ�������������в�����ȡ����Ķ��ٶ��仯����_______��
A����Һ��HCl�����ʵ��� B����Һ��Ũ��
C����Һ��Cl-����Ŀ D����Һ���ܶ�
��3��������ƿ��ʹ�÷����У����в�������ȷ����____________
A��ʹ������ƿǰ�����Ƿ�©ˮ
B������ƿ��ˮϴ�������ô�����Һϴ��
C��������Һʱ����������ǹ��壬�ѳƺõĹ�����ֽ��С�ĵ�������ƿ�У�������ˮ���ӽ��̶���1��2cm�����ý�ͷ�ιܼ�����ˮ���̶��ߡ�
D��������Һʱ����������Һ�壬����Ͳȡ�����ò�����������������ƿ�У�������ˮ���̶���1��2cm�����ý�ͷ�ιܼ�����ˮ���̶��ߡ�
E���Ǻ�ƿ������ʳָ��סƿ������һֻ����סƿ�ף�������ƿ������ת��Σ�ҡ�ȡ�
��4��ijѧ����������Ũ���������ˮ����500 mL���ʵ���Ũ��Ϊ0.400 mol��L-1��ϡ���ᡣ
�ٸ�ѧ����Ҫ��ȡ________mL����Ũ����������ơ�������С�����1λ��
�������ƹ����У�����ʵ������������Ƶ�ϡ��������ʵ���Ũ���к�Ӱ�죿(���������ƫ����ƫС������Ӱ�족)��
a������Ͳ��ȡŨ����ʱ���ӹ۲찼Һ�档( )
b�����ݺ���ҡ�ȡ����ã�����Һ���½����ټ�����������ˮ��( )
��5���ټ����ͬѧ�ɹ�������0.400 mol��L-1�����ᣬ�����ø������кͺ�0.4 g NaOH��NaOH��Һ�����ͬѧ��ȡ________mL���ᡣ
�ڼ����ͬѧ�������Ƶ������кͺ�0.4 g NaOH��NaOH��Һ�����ֱȢ����������ƫС������ܵ�ԭ����________��
A��Ũ����ӷ���Ũ�Ȳ��� B��������Һʱ��δϴ���ձ�
C��������Һʱ����������ƿ�̶��� D����ˮʱ�����̶��ߣ��ý�ͷ�ι�����
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2014��ӱ�ʡ�߶������ο��Ի�ѧ�Ծ��������棩 ���ͣ�ʵ����
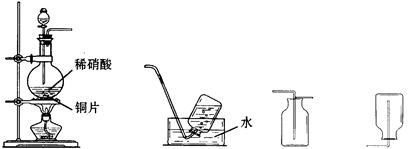
��11�֣�ijʵ��С���ͬѧΪ̽��һ�����������ʣ���ѡ������ʵ��װ������ȡһ��������
A B C D
��ش��������⣺
��1������ͼ����ʾ��װ����ȡһ��������ѡ���������ӵ�˳��Ӧ��A��_________��
��2��������15 mol/L HNO3��Һ����500 mL��1 mol/L HNO3��Һʱ��Ҫ�õ����������ձ�����ͷ�ιܡ�����������Ͳ�⣬������ʹ�õ�һ�ֲ��������� ��
��3��ϡ�������ǿ�����ԣ��ڳ����¾Ϳ��Ժ�ͭ������Ӧ����һ���������壬д���÷�Ӧ�Ļ�ѧ����ʽ��___________________________________________________��
��4��ʵ�鿪ʼʱ����С��ͬѧ��������ƿ���к���ɫ��������������û�ѧ����ʽ����ʾ�������������ԭ��_________________________________________________��
��5��һ��������һ����̼������β���е���Ҫ��Ⱦ�Ŀǰ������β���ѳ�Ϊ�������п�������Ҫ��ȾԴ����������β���е�NO��CO��һ�ַ����ǣ�����������������װһ����ת��װ�ã�ʹNO��CO��Ӧ������CO2��N2����Ӧ�Ļ�ѧ����ʽ�� ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2015�����ʡ��һ��ѧ�ڵ�һ���¿���ѧ�Ծ��������棩 ���ͣ�ʵ����
��14�֣�ʵ����������0.5 mol/L��NaOH��Һ500 ml���������������ձ�����100 ml��Ͳ����100 ml����ƿ����500 ml����ƿ���ݲ���������������ƽ(������)��
��1������ʱ������ʹ�õ������� ������ţ�����ȱ�ٵ������� ���������������õ��������������÷ֱ��� �� ��
��2��ʹ������ƿǰ������е�һ�������� ��
��3��������Һʱ��һ����Է�Ϊ���¼������裺�ٳ������ڼ��㣻���ܽ⣻��ҡ�ȣ���ת�ƣ���ϴ�ӣ��߶��ݣ�����ȴ������ȷ�IJ���˳��Ϊ ��
��4�������ƹ�����������������ȷ�ģ����в������������ƫ�ߵ��� ��
��û��ϴ���ձ��Ͳ����� ��δ��NaOH��Һ��ȴ�����¾�ת�Ƶ�������
������ƿ�����������������ˮ �ܶ���ʱ���ӱ��� �ݶ���ʱ���ӱ���
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2011-2012ѧ��ɽ��ʡ����10�·��¿���ѧ�Ծ� ���ͣ�ʵ����
(10��)Ϊά������ѪҺ�е�Ѫ�Ǻ������ڸ�������Һʱ��ͨ����������ע��Һ����ͼ��ҽԺ��������Һʱʹ�õ�һƿ��������Ϊ5%��������(C6H12O6)ע��Һ�ı�ǩ��ijѧ������ʵ����������500 mL��������ע��Һ��
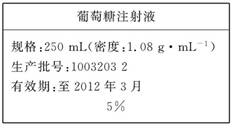
ʵ����Ʒ�������Ǿ���(Ħ��������180 g/mol)������ˮ���ձ�������ƿ(500 mL)��ҩ�ס���ͷ�ιܡ���Ͳ��
(1)��ȱ�ٵ�������_ ��
(2)���ж�����ƿ����ʹ�÷�������������ȷ����_ _��
A��ʹ��ǰҪ�������ƿ�Ƿ�©ˮ
B������ƿ������ˮϴ�������ñ�������ע��Һ��ϴ
C��������Һʱ���ѳƺõ������Ǿ���С�ĵ�������ƿ�У���������ˮ���ӽ��̶���1��2 cm�������ý�ͷ�ιܼ�����ˮ���̶���
D������ƿ�ϱ����ݻ����¶Ⱥ�Ũ��
(3)ʵ������ȡ�þ��������Ϊ__ __�����ʵ���Ũ��_ _��
(4)����0.1 mol/L��NaCl��Һ�����в����ᵼ��������Һ�����ʵ���Ũ��ƫ�ߵ���__ ��
A��û�н�ϴ��Һת�Ƶ�����ƿ B������ʱ���Ӷ�ȡ�̶�
C��ԭ����ƿϴ����δ���� D������ʱҺ�泬���˿̶���
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2010���㽭ʡ�߶��ڶ�ѧ�����п��ԣ�1-3�ࣩ��ѧ���� ���ͣ������
��19�֣�����������̵���Ҫ������ͳ��õ���������������ʵ������ģ�ҵ�������̿��Ʊ�������ص�����ͼ��
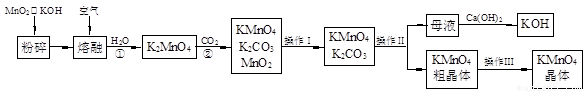
��1�������������Ϊ �������������Ϊ ��
��2����Ӧ�ڵĻ�ѧ����ʽΪ ����ҵ��������ԭ������KMnO4�������ʽϵͣ��Ϻõ��Ʊ������ǵ�ⷨ����Pt��������Fe��������K2MnO4Ϊ���Һ�������ĵ缫��ӦʽΪ ��
��3��KMnO4��һ�ֽ��ȶ��Ļ�������չ��KMnO4��Һ�ķֽ��д����ã�����MnO2��KOH��O2���� MnO2Ҳ�Ǹ÷ֽⷴӦ��һ�ִ������������һ��ʵ�鷽������֤MnO2�Ը÷ֽⷴӦ���д��ԣ� ��
��4��KMnO4�����Խ����е�ǿ�����Թ㷺Ӧ���ڷ�����ѧ�С�
���磺2KMnO4+3H2SO4+5Na2SO35Na2SO4+K2SO4+2MnSO4+3H2O��ijͬѧ��KMnO4�ⶨʵ���ҳ��ڴ�ŵ�Na2SO3����Ĵ��ȡ�����ȷ��ȡ6.3 g Na2SO3������Ʒ�����500 mL��Һ��ȡ25.00 mL������Һ������ƿ�У���0.01000 mol/L ������KMnO4��Һ���еζ����ζ�������±���ʾ��
|
�ζ�����[��Դ:][��Դ:Z&xx&k.Com] |
������Һ�����/mL[��Դ:ѧ#��#��Z#X#X#K] |
����Һ�����[��Դ:] |
|
|
�ζ�ǰ�̶�/mL |
�ζ���̶�/mL |
||
|
1 |
25.00 mL |
0.02 |
24.01 |
|
2 |
25.00 mL |
0.70 |
24.71 |
|
3 |
25.00 mL |
0.20 |
24.20 |
������500 mLNa2SO3��Һʱ�������õ���ʵ�������У��ձ�������������ͷ�ιܡ�ҩ�� �� ��
���жϵζ��յ�������� ��
�����в����ᵼ�²ⶨ���ƫ�ߵ���
A��δ�ñ�Ũ�ȵ�����KMnO4��Һ��ϴ�ζ���
B���ζ�ǰ��ƿδ����
C���ζ�ǰ�ζ��ܼ��첿��������
D���۲����ʱ���ζ�ǰ���ӣ��ζ�����
��������ʵ�����ݣ�����Na2SO3�Ĵ���Ϊ ��
�鿴�𰸺ͽ���>>
����ѧУ��ѡ - ��ϰ���б� - �����б�
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com