
�����й���Ԫ�ؼ��仯�����˵����������ȷ����(����)
A����ƿ��Ʊ����ᾭ��������S SO3
SO3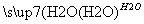 H2SO4
H2SO4
B�������������еĽ��������ﷴӦ����Ԫ���Ե��ʵ���ʽ����������
C����ȼú�м���ʯ��ʯ�ɼ���SO2�ŷţ������ķ�ӦΪ2CaCO3��2SO2��O2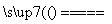 2CO2��2CaSO4
2CO2��2CaSO4
D�������е���п��(ZnS)��������ͭ��Һת��Ϊͭ��(CuS)��˵��CuS���ȶ��������л�ԭ��

| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
��������Ԫ�ػ�̬ԭ�ӵĵ����Ų������ж�Ԫ�أ����ش����⡣
A��ԭ����ֻ��һ�����Ӳ���ֻ��1�����ӣ�B��ԭ��3p����ϵõ�1�����Ӻ����������������ӣ�C��ԭ�ӵ�2p�������1�����ӵ������������������ӵ����������෴��D��ԭ�ӵ������Ӳ�����8�����ӣ����ĵ��Ӳ���ֻ��1�����ӣ�Eԭ�ӵ���Χ�����Ų�ʽΪ3s23p6��
(1)д��A��B��C��D����Ԫ�ص�Ԫ�ط��ţ�________________________________��
(2)����ij��Һ���Ƿ���D������ͨ��________��Ӧ��ʵ�֣�����ij��Һ���Ƿ���B����ͨ�����õ��Լ���__________��__________��
(3)д��E��Ԫ�ط���________��Ҫ֤��̫�����Ƿ���EԪ�أ��ɲ��õķ�����______��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
����˵����ȷ����(����)
A����ΪSO2����Ư���ԣ���������ʹƷ����Һ����ˮ������KMnO4��Һ��ʯ����Һ��ɫ
B����ʹƷ����Һ��ɫ�����ʲ�һ����SO2
C��SO2��Ư�ۡ�����̿��Na2O2����ʹ��īˮ��ɫ����ԭ��������ͬ
D�������ʵ�����SO2��Cl2��Ϻ�ͨ��װ��ʪ�����ɫ�����ļ���ƿ�У�Ư��Ч������
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
���������(Na2S2O3)�����������Լ�������Ļ�ԭ���������ȡ������ֽ⡣��ҵ�Ͽ��÷�Ӧ��2Na2S��Na2CO3��4SO2===3Na2S2O3��CO2 �Ƶá�ʵ����ģ��ù�ҵ���̵�װ����ͼ��ʾ���ش��������⣺
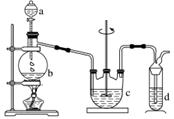
(1)b�з�Ӧ�����ӷ���ʽΪ_______________________________________________________��
c���Լ�Ϊ____________��
(2)��Ӧ��ʼ��c�����л��Dz��������ֱ���塣�˻�������____________��
(3)d�е��Լ�Ϊ______________��
(4)ʵ����Ҫ����SO2�������ʣ����Բ�ȡ�Ĵ�ʩ��
________________________________________________________________________
__________________________________________________________(�����)��
(5)Ϊ�˱�֤��������ƵIJ�����ʵ����ͨ��SO2���ܹ�����ԭ����________________________________________________________________________
________________________________________________________________________��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
�����������һ����Ҫ�Ļ�����Ʒ��ij��ȤС�����Ʊ���������ƾ���(Na2S2O3·5H2O)��
��.[��������]
(1)Na2S2O3·5H2O����ɫ�����壬������ˮ����ϡ��Һ��BaCl2��Һ����������ɡ�
(2)��Na2CO3��Na2S�����Һ��ͨ��SO2���Ƶ�Na2S2O3�����ò�Ʒ����������Na2SO3��Na2SO4��
(3)Na2SO3�ױ�������BaSO3������ˮ��������ϡHCl��
��.[�Ʊ���Ʒ]
ʵ��װ����ͼ��ʾ(ʡ�Լг�װ��)��
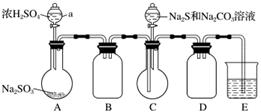
ʵ�鲽�裺
(1)���װ�������ԣ���ͼʾ�����Լ���
����a��������________��E�е��Լ���________(ѡ��������ĸ���)��
A��ϡH2SO4
B��NaOH��Һ
C������NaHSO3��Һ
(2)����C����ƿ����Na2S��Na2CO3�����Һ������A����ƿ�μ�ŨH2SO4��
(3)��Na2S��Na2CO3��ȫ���ĺ�����Ӧ������C�л�����Һ��__________(��д��������)���ᾧ�����ˡ�ϴ�ӡ�����õ���Ʒ��
��.[̽���뷴˼]
(1)Ϊ��֤��Ʒ�к���Na2SO3��Na2SO4����С�����������ʵ�鷽�����뽫��������������
(�����Լ���ϡHNO3��ϡH2SO4��ϡHCl������ˮ��ѡ��)
ȡ������Ʒ���ϡ��Һ���μ�����BaCl2��Һ���а�ɫ�������ɣ�________________��������δ��ȫ�ܽ⣬���д̼�����ζ��������������ȷ����Ʒ�к���Na2SO3��Na2SO4��
(2)Ϊ����װ��C������Na2SO4�������ڲ��ı�ԭ��װ�õĻ����϶�ʵ�鲽��(2)�����˸Ľ����Ľ���IJ�����
________________________________________________________________________��
(3)Na2S2O3·5H2O���ܽ�����¶����������������ò�Ʒͨ��________________�����ᴿ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
п��100 mL 18.5 mol·L��1��Ũ�����ַ�Ӧ��п��ȫ�ܽ⣬ͬʱ���������33.6 L(��״��)������Ӧ�����Һϡ����1 L�������Һ��pH��1��������������ȷ����(����)
A����Ӧ�й�����1.8 mol H2SO4
B���������SO2��H2�������Ϊ4��1
C����Ӧ�й�����97.5 g Zn
D����Ӧ�й�ת��1.5 mol����
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
ijԪ�ص�һ��ͬλ��X��ԭ��������ΪA����N�����ӣ�����1Hԭ�����HmX���ӣ���a g HmX���������ӵ����ʵ����� (����)
A.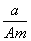 (A��N��m)mol B.
(A��N��m)mol B.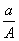 (A��N)mol
(A��N)mol
C.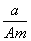 (A��N)mol D.
(A��N)mol D.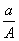 (A��N��m)mol
(A��N��m)mol
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
��ˮ(D2O)����Ҫ�ĺ˹�ҵԭ�ϣ�����˵���������(����)
A���(D)ԭ�Ӻ�����1������
B��1H��D����ͬλ��
C��H2O��D2O����ͬ��������
D��1H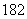 O��D
O��D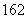 O����Է���������ͬ
O����Է���������ͬ
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
ʵ����ģ��ϳɰ��Ͱ����������������£�


��֪ʵ���ҿ��ñ�����������(NaNO2)��Һ�뱥���Ȼ����Һ�����Ⱥ�Ӧ��ȡ������
(1)��ͼ��ѡ����ȡ����ĺ���װ�ã�����______________������______________��
(2)����������ͨ����װ�ã���װ�õ����ó��˽��������⣬����________��_________��
(3)���ϳ�����������ȴ����������ͨ����װ�õ�ˮ�����հ���________(���ᡱ���ᡱ)����������ԭ����______________________________________________________��
(4)����װ������һ��ʱ�䰱����ͨ�������ͬʱ�������ȵIJ�˿������װ�õ���ƿ�ڣ���ʹ��˿���ֺ��ȵ�ԭ����____________________________________________��
��ƿ�л��ɹ۲쵽��������________________________________________________��
(5)д����װ���а������Ļ�ѧ����ʽ��____________________________________
________________________________________________________________________��
(6)��Ӧ��������ƿ�ڵ���Һ�к��е�����ΪH����OH����________��________��
�鿴�𰸺ͽ���>>
����ѧУ��ѡ - ��ϰ���б� - �����б�
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com