
| ��һ�� | �ڶ��� | ������ | |
| �����mL�� | 16.10mL | 16.60mL | 15.90mL |
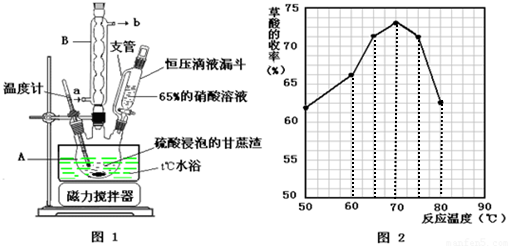
���� ��1���������ṹ��������֪����AΪ������ƿ��
��2�����ݲ���Ļ��������ʱ�ж���ѷ�Ӧ�¶ȣ�Ũ���ᡢ�������ǿ�����ԣ������������
��3�������������Ҵ���ˮ���������ѣ��������ӷ���������ϴ�ӣ�
��4�������������¸�����ؽ���������Ϊ������̼����������ԭΪ�����ӣ�
��A�����Ʊ�Һʱ����KMnO4���������ʲ������Һ��Ӧ�������ĸ��������Һ���ƫ��
B����ϴ�ζ���ʱ������Ҫ�������������Һ������ҩƷ���˷ѣ�
C���ζ�ʱ���۾�ע����ƿ���۲���Һ��ɫ�仯���жϷ�Ӧ�յ㣻
D���ζ��յ��ȡ�ζ��̶ܿ�ʱ�����ӱ�ҺҺ�棬��ʹ���ĸ��������Һ���������ƫ��
���������Ը��������Һ���Ϻ�ɫ�����ᷴӦ��ϣ��������һ�θ�����ر�Һ����ƿ����Һ���dz�죬��Ӧ�����յ㣻
�ܵ�2�������������������ϴ�Ӧ��������������ƽ��ֵΪ�������Ը��������Һ���������n=cV���㷴Ӧ�����ĸ�����ص����ʵ��������ݷ���ʽ5H2C2O4+2MnO4-+6H+=2Mn2++10CO2��+8H2O����25mL��������ʵ������ټ���ԭ��Ʒ�в�������ʵ���������������ᾧ��Ĵ��ȣ�
��� �⣺��1���������ṹ��������֪����AΪ������ƿ��
�ʴ�Ϊ��������ƿ��
��2������ͼ3��֪�����¶�Ϊ70��ʱ�������������ߣ�����ѡ�����ѷ�Ӧ�¶���70�棬
Ũ���ᡢ�������ǿ�����ԣ������������ᣬ����μӹ�����¶ȹ��ߣ������²�������½���
�ʴ�Ϊ��70�棻�����ױ�Ũ���������������
��3�������������Ҵ���ˮ���������ѣ��������ӷ���������ϴ�ӣ�
�ʴ�Ϊ�����ѣ��ӷ���������
��4�������������¸�����ؽ���������Ϊ������̼����������ԭΪ�����ӣ���Ӧ���ӷ���ʽΪ��5H2C2O4+2MnO4-+6H+=2Mn2++10CO2��+8H2O��
�ʴ�Ϊ��5H2C2O4+2MnO4-+6H+=2Mn2++10CO2��+8H2O��
���ݲ�����Һ������Լ���ȡ��Һ�ľ�ȷ��ѡ����Ӧ��������
��A�����Ʊ�Һʱ����KMnO4���������ʲ������Һ��Ӧ�������ĸ��������Һ���ƫ��ʹʵ�����ƫƫ�ߣ���A����
B����ϴ�ζ���ʱ������Ҫ�������������Һ������ҩƷ���˷ѣ���B����
C���ζ�ʱ���۾�ע����ƿ���۲���Һ��ɫ�仯���жϷ�Ӧ�յ㣬��C��ȷ��
D���ζ��յ��ȡ�ζ��̶ܿ�ʱ�����ӱ�ҺҺ�棬��ʹ���ĸ��������Һ���������ƫ��ʹʵ�����ƫ�ߣ���D��ȷ��
��ѡ��CD��
���������Ը��������Һ���Ϻ�ɫ�����Եζ��յ���ж������ǣ�����ƿ�е������һ�θ�����ر�Һ����ƿ����Һ���dz��ɫ�Ұ���Ӻ�ɫ��
�ʴ�Ϊ������ƿ�е������һ�θ�����ر�Һ����ƿ����Һ���dz��ɫ�Ұ���Ӻ�ɫ��
�ܵ�2�������������������ϴ�Ӧ�������������Ը��������Һ���Ϊ$\frac{��16.10+15.90��ml}{2}$=16.00mL����Ӧ�����ĸ�����ص����ʵ�����0.016L��0.1mol/L=0.0016mol�����ݷ���ʽ��5H2C2O4+2MnO4-+6H+=2Mn2++10CO2��+8H2O����֪25mL��Һ�в�������ʵ�����0.0016mol��$\frac{5}{2}$=0.004mol�����ԭ��Ʒ�в�������ʵ�����0.004mol��$\frac{250ml}{25ml}$=0.04mol��������=0.04mol��126g/mol=5.04g�����Բ���Ĵ�����$\frac{5.04g}{10.0g}$��100%=50.4%��
�ʴ�Ϊ��50.4%��
���� ���⿼���л����Ʊ�ʵ�鷽����ƣ��漰��ѧ��������װ�õķ������ۡ�ʵ��������ѡ����ơ����ʺ����IJⶨ��������ԭ��Ӧ�ζ��ȣ��Ƕ�ѧ���ۺ������Ŀ��飬��Ҫѧ���߱���ʵ�Ļ�������Ŀ�Ѷ��еȣ�


 ������ȫ��������ϵ�д�
������ȫ��������ϵ�д�
| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ѡ����
| A�� | ���ʷ�����ѧ��Ӧ�������������ı仯�����������仯�����ʱ仯���ǻ�ѧ�仯 | |
| B�� | 101kPaʱ��2H2��g��+O2��g���T2H2O��l����H=-571.6kJ•mol-1��H2��g����ȼ����Ϊ285.8kJ•mol-1 | |
| C�� | ����ͬ�����µ�ϡ��Һ�У�1molHCl��1molHNO3�ֱ�������NaOH��ַ�Ӧ���ų���������� | |
| D�� | H2+Cl2�T2HCl�������仯���������ͼ��ʾ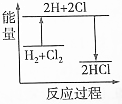 |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ѡ����
| A�� | �� | B�� | �٢ڢ� | C�� | �٢ڢܢ� | D�� | �٢ڢ� |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������

| ��� | �۵�/�� | �е�/�� | �ܶȣ�20�棩/g•cm-3 | �ܽ��� |
| �� | 5.5 | 80 | 0.88 | ����ˮ |
| ������ | 5.7 | 210.9 | 1.205 | ������ˮ |
| Ũ���� | - | 83 | 1.4 | ������ˮ |
| Ũ���� | - | 338 | 1.84 | ������ˮ |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ʵ����
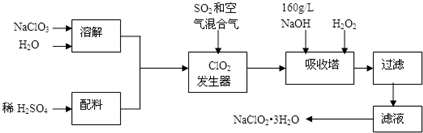
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
 A��ʯ���ѽ���Ҫ����֮һ������������ں���һ��ʯ�ͻ�����չˮƽ�ı�־��
A��ʯ���ѽ���Ҫ����֮һ������������ں���һ��ʯ�ͻ�����չˮƽ�ı�־���鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ѡ����
| A�� | ��pH=1����Һ�У�SO32-��Cl-��NO3-��Na+ | |
| B�� | ����ʹ��̪������Һ�У�Na+��Cl-��NO3-��K+ | |
| C�� | ��1 mol•L-1��NaAlO2��Һ�У�K+��Ba2+��SO42-��OH- | |
| D�� | ��1 mol•L-1��AlCl3��Һ�У�NH4+��Ag+��Na+��NO3- |
�鿴�𰸺ͽ���>>
����ѧУ��ѡ - ��ϰ���б� - �����б�
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com