
��֪��H2(g)��F2(g)===2HF(g) ��H����270 kJ·mol��1������˵����ȷ����(����)
A��2 L����������ֽ��1 L������1 L��������270 kJ����
B��1 mol������1 mol������Ӧ����2 molҺ̬������ų�������С��270 kJ
C������ͬ�����£�1 mol������1 mol�����������ܺʹ���2 mol���������������
D��1������������1���������ӷ�Ӧ����2��������������ӷų�270 kJ����
 ��У����ϵ�д�
��У����ϵ�д�
| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
������������ͬ��ͭ����п�����õ��������Ӻ����CuSO4��Һ�У�����һ��ʱ���ȡ��ϴ������ﲢ���أ������������6.45 g��������ͨ���ĵ��ӵ����ʵ���Ϊ(����)
A��0.1 mol B��0.2 mol
C��0.3 mol D��0.4 mol
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
Ϊ��ȷ���л������Ƿ���ڵ����ȡ��塢���Ԫ�أ�Ӧ���л�����Ʒ���������ֽ���������ں��������Է�������
A�� Fe B�� Si C�� Cu D�� Na
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
����˵������ȷ���ǣ���
A�� ����ʽΪC3H8��C6H14�������л���һ����Ϊͬϵ��
B�� ������ͬͨʽ���л��ﲻһ����Ϊͬϵ��
C�� ��������ͬϵ�����Է���������ֵһ�����14
D�� ����������һ�������ɸ�CH2ԭ���ŵĻ�����ض���Ϊͬϵ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
�л���A��̼���⡢������Ԫ����ɣ���ȡ3g A��4.48L��������״�������ܱ�������ȼ�գ�ȼ�պ����ɶ�����̼��һ����̼��ˮ���������跴Ӧ��û��ʣ�ࣩ������Ӧ���ɵ���������ͨ��Ũ����ͼ�ʯ�ң�Ũ��������3.6g����ʯ������4.4g���ش��������⣺
��1��3g A��������ԭ�ӡ�̼ԭ�ӵ����ʵ������Ƕ��٣�
��2��ͨ������ȷ�����л���ķ���ʽ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�

����ͼ��ʾ�����Թܷ���ʢ��25 ��ı���ʯ��ˮ���ձ��У��Թ��п�ʼ���뼸С��þƬ�����õιܵμ�5 mL�������Թ��С��Իش��������⣺
(1)ʵ���й۲쵽��������______________________________��
(2)�������������ԭ����______________________________��
(3)д���йط�Ӧ�����ӷ���ʽ��_______________________��
(4)��ʵ����֪��þƬ�������������________(����ڡ���С�ڡ����ڡ�)�Ȼ�þ��Һ����������������
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
��ѧ��Ӧ���������ʲ�����ͬʱ�������������仯��
(1)��һ��С�ձ������Լ20 g����ĥ�ɷ�ĩ��������������[Ba(OH)2·8H2O]����С�ձ����������ѵ���3��4��ˮ�IJ���Ƭ�ϣ�Ȼ���ټ���Լ10 g NH4Cl���壬�������ò�����Ѹ�ٽ���(����ͼ)��
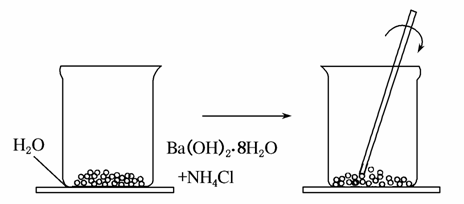
��ش��������⣺
�ٲ����У������ò�����Ѹ�ٽ����Ŀ����______________��
�ڷ�����Ӧ�Ļ�ѧ����ʽΪ____________________________��
�۹۲쵽������(�д̼�����ζ����)��_____________________ _________���������˵����������__________________________��
(2)ij��ѧС�黹�����һ��ʵ��(��ͼ)��
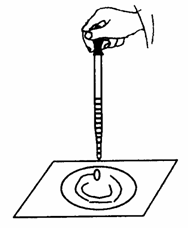
����֬�ް�סԼ0.2 g Na2O2��ĩ������ʯ�����ϡ�����֬���ϵμӼ���ˮ��
��ش��������⣺
�ٹ���������ˮ������Ӧ�Ļ�ѧ����ʽΪ_______________________________________________________��
�ڹ۲쵽��������__________________������������������ԭ��_________________________________________________________��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
һ�������·�ӦmA��g��+nB��g��⇌pC��g��+qD��g����һ�ܱ������н��У����ƽ����Ӧ���ʦԣ�C��=2�ԣ�B�����ҷ�Ӧǰ�������ڵ�ѹǿ���䣬��m��n��p��q����ֵ�ǣ�������
| �� | A�� | 2��1��2��1 | B�� | 3��1��2��2 | C�� | 2��6��3��5 | D�� | 1��3��2��2 |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2014-2015����ʡ�����и߶���ѧ����ĩ���Ի�ѧ�Ծ��������棩 ���ͣ������
(12��)�����£���ijһԪ��HA��NaOH��Һ�������ϣ�������Һ��Ũ�Ⱥͻ�Ϻ�������Һ��pH���±���
ʵ�� ��� | HA���ʵ��� Ũ��(mol��L��1) | NaOH���ʵ��� Ũ��(mol��L��1) | ����� Һ��pH |
�� | 0.1 | 0.1 | pH��9 |
�� | c | 0.2 | pH��7 |
�� | 0.2 | 0.1 | pH<7 |
��ش�
��1���Ӣ���ʵ�������HA��________(�ǿ�ᡱ�����ᡱ)��
��2������ʵ�������c________0.2(����ڡ�����С�ڡ����ڡ�)�����Һ������Ũ��c(A��)��c(Na��)�Ĵ�С��ϵ��__________________��
��3���Ӣ���ʵ����������˵��HA�ĵ���̶�______(����ڡ�����С�ڡ����ڡ�)NaA��ˮ��̶ȣ��û����Һ������Ũ���ɴ�С��˳����_______________________��
��4������ʵ�����õĻ����Һ�У�������ʽ�ľ�ȷ����ǣ�c(OH-)- c(HA)=_________mol/L
�鿴�𰸺ͽ���>>
����ѧУ��ѡ - ��ϰ���б� - �����б�
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com