
���к�FeC12���ʵ��Ȼ�ͭ���壨CuC12?2H2O����Ϊ��ȡ������CuC12?2H2O�����Ƚ����Ƴ�ˮ��Һ��Ȼ����ͼ��ʾ��������ᴿ��
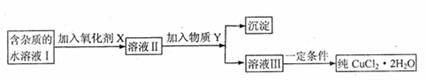
��֪H2O2��KMnO2��NaC1O��K2Cr2O7������ǿ�����ԣ�Ҫ����Һ�е�Cu2+��Fe2+��Fe3+����Ϊ�����������Һ��pH�ֱ�Ϊ6.4��6.4��3.7��
��ش���������
��1����ʵ�����ʺϵ�������X�� ��
A��K2Cr2O7 B��NaC1O C��H2O2 D��KMnO4
��2������Y�������� ��
A��CuO B��CuC12 C��Cu��OH��2 D��CuCO3
��3����ȥFe3+���й����ӷ���ʽ�ǣ�����Y�ڣ�2����ѡ����ʵ�ѡ��� ��
��4��������������Ŀ���� ��
��5������ܲ���ֱ�������ᾧ�õ�CuC12?2H2O�� ������ܡ����ܡ������粻�ܣ�Ӧ��β����������ܣ��˿ղ�� ��

| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
����ѧУ��ѡ - ��ϰ���б� - �����б�
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com