
(12��)ij��Ԫ����(��дΪH2A)��Һ������ʽ����һ���Ͷ������룺H2AH����HA���� HA��
H����A2 ����֪��ͬŨ��ʱ�ĵ���Ȧ�(H2A)����(HA��)����������������Һ��
A. 0.01 mol��L��1��H2A��Һ
B. 0.01 mol��L��1��NaHA��Һ
C.0.02 mol��L��1��HCl��0.04mol��L��1��NaHA��Һ��������Һ
D.0.02 mol��L��1��NaOH��0.02mol��L��1��NaHA��Һ��������Һ,�ݴˣ���д���пհ�(�����)��
(1)c(H��)������ ,������ ��
(2)c(H2A)������ ,������ ��
(3)c(A2��)������ ,������ ��

| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ��2013�������ʡ˫Ѽɽ�е�һ��ѧ�����������¿���ѧ�Ծ����������� ���ͣ������
(12��) A��B��W��D��EΪ������Ԫ�أ���ԭ��������������������֮��Ϊ39��B��Wͬ���ڣ�A��Dͬ���壬A��W���γ�����Һ̬������A2W��A2W2��EԪ�ص���������������������ȡ�
��1��EԪ�������ڱ��е�λ��Ϊ ��
��2����A��B��W����Ԫ����ɵ�18�������ĵ���ʽΪ ��
��3�����ⶨA2W2Ϊ��Ԫ���ᣬ�����Ա�̼��Ļ�Ҫ������д�����һ������ĵ��뷽��ʽ ���������ᴦ��BaO2���Ʊ�A2W2��д���÷�Ӧ�Ļ�ѧ����ʽ ��
��4����ӡˢ��·���Ϻ���ͭ�������Ļ��շ����ǽ�������ʹͭת��Ϊ����ͭ�����������ܽ⡣�ָ���A2W2��ϡ������ݷ�ӡˢ��·��ȴﵽ����Ŀ�ģ��ֱ����˻�������д����Ӧ�����ӷ���ʽ ��
��5��Ԫ��D�ĵ�����һ�������£�����A���ʻ�������һ���Ȼ���DA���۵�Ϊ800��DA����ˮ��Ӧ������������1molDA��1molE���ʻ�ϼ���������ˮ����ַ�Ӧ�����ȫ������� ����״���£���
��6��D��ij������ʵ���ɫ�������Ȼ�������Һ��Ӧ��������ɫ�������Ȼ�������Ӧ�����ʵ���֮��Ϊ1��2�������������ɣ���÷�Ӧ�����ӷ���ʽΪ ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2011-2012ѧ������ʡ�����е�һ��ѧ�߶���ѧ����ĩ���Ի�ѧ�������Ծ� ���ͣ������
(12��)ij��Ԫ����(��дΪH2A)��Һ������ʽ����һ���Ͷ������룺 H2A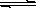 H����HA���� HA��
H����HA���� HA��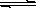 H����A2 ����֪��ͬŨ��ʱ�ĵ���Ȧ�(H2A)����(HA��)����������������Һ��
H����A2 ����֪��ͬŨ��ʱ�ĵ���Ȧ�(H2A)����(HA��)����������������Һ��
A. 0.01 mol��L��1��H2A��Һ
B. 0.01 mol��L��1��NaHA��Һ
C.0.02 mol��L��1��HCl��0.04 mol��L��1��NaHA��Һ��������Һ
D.0.02 mol��L��1��NaOH��0.02 mol��L��1��NaHA��Һ��������Һ,�ݴˣ���д���пհ�(�����)��
(1)c(H��)������ ,������ ��
(2)c(H2A)������ ,������ ��
(3)c(A2��)������ ,������ ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2012-2013ѧ�������ʡ˫Ѽɽ�и����������¿���ѧ�Ծ��������棩 ���ͣ������
(12��) A��B��W��D��EΪ������Ԫ�أ���ԭ��������������������֮��Ϊ39��B��Wͬ���ڣ�A��Dͬ���壬A��W���γ�����Һ̬������A2W��A2W2��EԪ�ص���������������������ȡ�
��1��EԪ�������ڱ��е�λ��Ϊ ��
��2����A��B��W����Ԫ����ɵ�18�������ĵ���ʽΪ ��
��3�����ⶨA2W2Ϊ��Ԫ���ᣬ�����Ա�̼��Ļ�Ҫ������д�����һ������ĵ��뷽��ʽ ���������ᴦ��BaO2���Ʊ�A2W2��д���÷�Ӧ�Ļ�ѧ����ʽ ��
��4����ӡˢ��·���Ϻ���ͭ�������Ļ��շ����ǽ�������ʹͭת��Ϊ����ͭ�����������ܽ⡣�ָ���A2W2��ϡ������ݷ�ӡˢ��·��ȴﵽ����Ŀ�ģ��ֱ����˻�������д����Ӧ�����ӷ���ʽ ��
��5��Ԫ��D�ĵ�����һ�������£�����A���ʻ�������һ���Ȼ���DA���۵�Ϊ800��DA����ˮ��Ӧ������������1molDA��1molE���ʻ�ϼ���������ˮ����ַ�Ӧ�����ȫ������� ����״���£���
��6��D��ij������ʵ���ɫ�������Ȼ�������Һ��Ӧ��������ɫ�������Ȼ�������Ӧ�����ʵ���֮��Ϊ1��2�������������ɣ���÷�Ӧ�����ӷ���ʽΪ ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2013������ʡ�����и߶���ѧ����ĩ���Ի�ѧ�������Ծ� ���ͣ������
(12��)ij��Ԫ����(��дΪH2A)��Һ������ʽ����һ���Ͷ������룺
H2A H����HA����
HA��
H����HA����
HA�� H����A2 ����֪��ͬŨ��ʱ�ĵ���Ȧ�(H2A)����(HA��)����������������Һ��
H����A2 ����֪��ͬŨ��ʱ�ĵ���Ȧ�(H2A)����(HA��)����������������Һ��
A. 0.01 mol��L��1��H2A��Һ
B. 0.01 mol��L��1��NaHA��Һ
C.0.02 mol��L��1��HCl��0.04 mol��L��1��NaHA��Һ��������Һ
D.0.02 mol��L��1��NaOH��0.02 mol��L��1��NaHA��Һ��������Һ,�ݴˣ���д���пհ�(�����)��
(1)c(H��)������ ,������ ��
(2)c(H2A)������ ,������ ��
(3)c(A2��)������ ,������ ��
�鿴�𰸺ͽ���>>
����ѧУ��ѡ - ��ϰ���б� - �����б�
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com