
�ϳɰ��������ѧ�����ϵ�һ���ش�ͻ�ƣ��䷴Ӧԭ��Ϊ
N2(g)��3H2(g)2NH3(g)����H����92.4 kJ��mol��1��
һ�ֹ�ҵ�ϳɰ��ļ�ʽ����ͼ���£�

(1)��Ȼ���е�H2S���ʳ��ð�ˮ���գ�����ΪNH4HS��һ����������NH4HS��Һ��ͨ��������õ�������ʹ����Һ������д��������Ӧ�Ļ�ѧ����ʽ��________________________________________________________________________��
(2)���������������ԭ�����£�
��CH4(g)��H2O(g)CO(g)��3H2(g)
��H����206.4 kJ��mol��1
��CO(g)��H2O(g)CO2(g)��H2(g)
��H����41.2 kJ��mol��1
���ڷ�Ӧ�٣�һ���������ƽ����ϵ��H2�İٷֺ��������ܼӿ췴Ӧ���ʵĴ�ʩ��____________��
a�������¶ȡ�b������ˮ����Ũ�ȡ�c�����������d������ѹǿ
���÷�Ӧ�ڣ���CO��һ��ת���������H2�IJ�������1 mol CO��H2�Ļ������(CO���������Ϊ20%)��H2O��Ӧ���õ�1.18 mol CO��CO2��H2�Ļ�����壬��CO��ת����Ϊ____________��
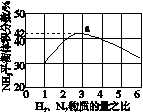
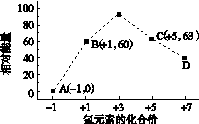
(3)ͼ(a)��ʾ500 �桢60.0 MPa�����£�ԭ����Ͷ�ϱ���ƽ��ʱNH3��������Ĺ�ϵ������ͼ��a�����ݼ���N2��ƽ�����������____________��
(4)�����¶ȶԺϳɰ���Ӧ��Ӱ�죬��ͼ(b)����ϵ�У�����һ�������µ��ܱ������ڣ���ͨ��ԭ������ʼ�����¶Ȳ������ߣ�NH3���ʵ����仯������ʾ��ͼ��
 ��
��
(a)������������������(b)
(5)��������ͼ�У�ʹ�ϳɰ��ų��������õ�������õ���Ҫ������(�����)________����������������ߺϳɰ�ԭ����ת���ʵķ�����________________________________________________________________________
________________________________________________________________________��
(1)2NH4HS��O2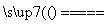 2NH3��H2O��2S��
2NH3��H2O��2S��
(2)a��90%
(3)14.5%
(4)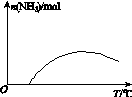
(5)������ԭ������ѹ������Һ����δ��Ӧ��N2��H2ѭ��ʹ��
[����] (1)�������֪Ϊ�����е�O2��������������Ϊ���ʣ����ݵ����غ㽫����ʽ��ƽ���ɡ�(2)��Ӧ��Ϊ�������ʵ�����������ȷ�Ӧ������ѹǿʹƽ�����ƣ�����Ӧ���ʼ�С��d�����������ܸı䷴Ӧ�ȣ������ܸı�H2�İٷֺ�����c��������ˮ����Ũ�����ʹ��Ӧ���������Լ�ƽ�����ƣ�������H2�İٷֺ���ȴ��С��b���������¶ȷ�Ӧ����������ƽ�������ƶ���H2�İٷֺ�������a�ԡ�CO��H2�Ļ��������ˮ�����ķ�Ӧ�У���Ӧ��ϵ�е���������ʵ������䣬��1 molCO��H2�Ļ������μӷ�Ӧ����1.18 mol�������˵����0.18 mol ˮ�����μӷ�Ӧ������ݷ���ʽ�ڿ�֪�μӷ�Ӧ��COҲΪ0.18 mol������ת����Ϊ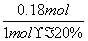 ��100%��90%��
��100%��90%��
(3)��ͼ�п�����N2��H2���ʵ�����Ϊ1��3ʱ��NH3��ƽ������������Ϊ42%����ƽ��ʱת����N2�����ʵ���Ϊx mol��������ʽ��
����������N2��3H2 2NH3
2NH3
��ʼ(mol): 1 3 0
ת��(mol): x 3x 2x
ƽ��(mol): 1��x 3�� 3x 2x
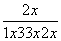 ��100%��42%����x��0.59
��100%��42%����x��0.59
��ƽ��ʱN2���������Ϊ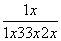 ��100%��14.5%��(4)��ͼʱҪע�ʼʱNH3���ʵ����������࣬����Ϊ��Ӧ�������(��Ӧδ��ƽ��)���ﵽһ���̶Ⱥ�Ӧ�ﵽƽ�����ʱ�¶ȼ������ߣ�ƽ�������ƶ���NH3�����ʵ�����С��(5)�Ƚ���������ʹ��Ҫ���ȵ����ʵõ����ȣ�������ʹ��Ҫ��ȴ�����ʵõ���ȴ���ܳ�������������ϳɰ���ӦΪ�������ʵ�����С�ķ�Ӧ����ѹ���ڷ�Ӧ������У����⣬ѭ�����ÿɷ�������ԭ�ϣ����ԭ�������ʡ�
��100%��14.5%��(4)��ͼʱҪע�ʼʱNH3���ʵ����������࣬����Ϊ��Ӧ�������(��Ӧδ��ƽ��)���ﵽһ���̶Ⱥ�Ӧ�ﵽƽ�����ʱ�¶ȼ������ߣ�ƽ�������ƶ���NH3�����ʵ�����С��(5)�Ƚ���������ʹ��Ҫ���ȵ����ʵõ����ȣ�������ʹ��Ҫ��ȴ�����ʵõ���ȴ���ܳ�������������ϳɰ���ӦΪ�������ʵ�����С�ķ�Ӧ����ѹ���ڷ�Ӧ������У����⣬ѭ�����ÿɷ�������ԭ�ϣ����ԭ�������ʡ�


 �����Ƹ���ʦ����ϵ�д�
�����Ƹ���ʦ����ϵ�д� ��ͨ����ͬ����ϰ��ϵ�д�
��ͨ����ͬ����ϰ��ϵ�д� ����С����ͬ������ϵ�д�
����С����ͬ������ϵ�д�
| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
b gij������������ϡ���ᷴӦ�����ɸý������������κ�a g�������ý��������ԭ������Ϊ(����)
A��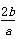
B��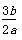
C��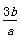
D��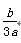
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
����ȷ��ʾ���з�Ӧ�����ӷ���ʽ����(����)
A��Cl2ͨ��NaOH��Һ��
Cl2��OH��===Cl����ClO����H2O
B��NaHCO3��Һ�м���ϡ���
CO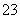 ��2H��===CO2����H2O
��2H��===CO2����H2O
C��AlCl3��Һ�м������ϡ��ˮ��
Al3����4NH3��H2O===AlO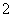 ��4NH
��4NH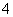 ��2H2O
��2H2O
D��Cu����ϡHNO3��
3Cu��8H����2NO ===3Cu2����2NO����4H2O
===3Cu2����2NO����4H2O
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
��̵�ص����С�������������dz��õ�һ�ε�ء��õ�ط�Ӧԭ����ͼ��ʾ�����е����LiClO4�����ڻ���л��ܼ��У�Li��ͨ�������Ǩ����MnO2�����У�����LiMnO2�� �ش��������⣺
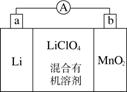
(1)���·�ĵ�����������________������________����(����ĸ)
(2)���������ӦʽΪ__________________________��
(3)�Ƿ����ˮ�������еĻ���л��ܼ���________(��ǡ���)��ԭ����________________________________________________��
(4)MnO2����KOH��KClO3�ڸ����·�Ӧ������K2MnO4����Ӧ�Ļ�ѧ����ʽΪ____________________________________��K2MnO4��������Һ���绯������KMnO2��MnO2�����ʵ���֮��Ϊ________��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
Ԫ�����ڱ��еڢ�A��Ԫ�صĵ��ʼ��仯�������;�㷺��
(1)����Ԫ��ͬ��Ķ�����Ԫ�ص�ԭ�ӽṹʾ��ͼΪ________��
(2)����Ϊ�ȡ��塢��Ԫ�طǽ�����(ԭ�ӵõ�������)�ݱ���ɵ��ж�������________(�����)��
a��Cl2��Br2��I2���۵�
b��Cl2��Br2��I2��������
c��HCl��HBr��HI�����ȶ���
d��HCl��HBr��HI������
(3)��ҵ�ϣ�ͨ������ת�����Ƶ�KClO3���壺
NaCl��Һ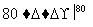 NaClO3��Һ
NaClO3��Һ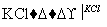 KClO3�������ɢ��з�Ӧ���ܻ�ѧ����ʽ��
KClO3�������ɢ��з�Ӧ���ܻ�ѧ����ʽ��
 NaCl��
NaCl�� H2O===
H2O=== NaClO3��
NaClO3�� ________��
________��
�ڢ���ת���Ļ�����Ӧ������________________���÷�Ӧ����������KClO3���������������������ԭ����____________________________________��
(4)һ�������£���ˮ��Һ��1 mol Cl����ClO (x��1��2��3��4)������(kJ)��Դ�С����ͼ��ʾ��
(x��1��2��3��4)������(kJ)��Դ�С����ͼ��ʾ��
��D��________(�����ӷ���)��
��B��A��C��Ӧ���Ȼ�ѧ����ʽΪ________________(�����ӷ��ű�ʾ)��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
�����йػ�ѧ�����ʾ��ȷ����(����)
A���������Ƶĵ���ʽ��
B��������Ϊ35��������Ϊ45����ԭ�ӣ� Br
Br
C�������ӵĽṹʾ��ͼ��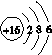
D������ױ��Ľṹ��ʽ��H3CCH3
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
SO2����������Fe2(SO4)3��Һ��ȫ��Ӧ���ټ���K2Cr2O7��Һ����������������ѧ��Ӧ��
��SO2��2Fe3����2H2O===SO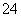 ��2Fe2����4H����
��2Fe2����4H����
��Cr2O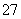 ��6Fe2����14H��===2Cr3����6Fe3����7H2O��
��6Fe2����14H��===2Cr3����6Fe3����7H2O��
�����й�˵���������(����)
A�������ԣ�Cr2O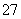 >Fe3��>SO2
>Fe3��>SO2
B��K2Cr2O7�ܽ�Na2SO3������Na2SO4
C��ÿ��1 mol K2Cr2O7�μӷ�Ӧ��ת�Ƶ��ӵ���ĿΪ6NA
D������6.72 L SO2(��״��)�μӷ�Ӧ������������0.2 mol K2Cr2O7
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
ʵ������ȡ��ϩ��װ����ͼ��ʾ���Իش�
(1)��ƿ�з�����Ӧ�Ļ�ѧ����ʽΪ_________________________
______________________________________________________________________________________________________________��
(2)ʵ���з����Թܢ�����ˮ��ɫ���������ܷ�˵��һ������ϩ����________(��ܡ����ܡ�)��
�Թ�����һ�����֤��ȷʵ����ϩ���ɣ���������__________���Թܢ�����ˮ��ɫ�ܷ�˵����������ϩ����ˮ�����˼ӳɷ�Ӧ________(��ܡ����ܡ�)��ͨ���ⶨ��Ӧ����Һ��pH�ɽ�һ��֤���Ƿ�����ȡ����Ӧ���Ǽӳɷ�Ӧ��ԭ����__________________
______________________________________________________________________________________________________________________��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
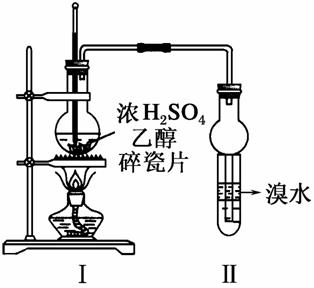
ij���ͭ�������ַ�ˮ��Ҫ������һ�ַ�ˮ�к���CN������һ�ַ�ˮ�к���Cr2O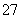 ���ó��ⶨ��ͼ��ʾ�ķ�ˮ�������̡�
���ó��ⶨ��ͼ��ʾ�ķ�ˮ�������̡�
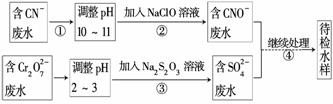
�ش��������⣺
(1)����������ˮ����������Ҫʹ�õķ�����___ _____________________________________________________________________��
_____________________________________________________________________��
(2)����ʹ�õ�NaClO��Һ�ʼ��ԣ������ӷ���ʽ����ԭ��________________________________________________________________________��
(3)���з�Ӧ��������ų����÷�Ӧ�����ӷ���ʽΪ
________________________________________________________________________��
(4)���з�Ӧʱ��ÿ0.4 mol Cr2O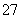 ת��2.4 mol e�����÷�Ӧ�����ӷ���ʽΪ________________________________________________________________________��
ת��2.4 mol e�����÷�Ӧ�����ӷ���ʽΪ________________________________________________________________________��
(5)ȡ��������ˮ�����Թ��У��ȼ���NaOH��Һ���۲쵽����ɫ�������ɣ���������NaOH��Һ��ֱ�����ٲ�����ɫ����Ϊֹ���ټ���Na2S��Һ���к�ɫ�������ɣ�����ɫ�������١�����ʹ�û�ѧ�����ϱ�Ҫ�����ֽ�����ԭ��________________________________________________________________________��
�鿴�𰸺ͽ���>>
����ѧУ��ѡ - ��ϰ���б� - �����б�
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com