
(1)��д��ͨʽΪCxHy��ij���ڹ�������ʱ��ȼ�յĻ�ѧ����ʽ(CO2��ϵ���趨Ϊm)��
(2)��m=2ʱ��������ķ���ʽ��
(3)��1 molij��̬���ڹ�������ʱȼ�գ�����������Na2O2�͵������������
����3 mol�������ҹ���Na2O2���ط�ΧΪ90g�ܦ�����118g���������ܵķ���ʽ��ȼ�ղ���CO��CO2�����ʵ���֮�ȣ�����������±�
���ķ���ʽ | n��CO��/n��CO2�� |
|
|
�𰸣�
��1��CxHy+![]() O2��mCO1+
O2��mCO1+![]() H2O+(x-m)CO
H2O+(x-m)CO
(2)

��m=2ʱ����ã�x=4 y=8 ���Ը���ΪC4H8
(3)
����ʽ |
|
C3H8 |
|
C4H6 |
|
���¼�������۹��̹��ο���
��������������
������������CO2��+��W��H2O��+��W��CO����28��+y+28(x-m)��28x+y
�ٸ���Na2O2���� 90��28x+y��118���۵���̬����x=3ʱy��6��x=4ʱ����6��
�������º����Ĵ��У�C4H6��C4H4��C3H8��C3H6
�ڸ���ȼ�ղ���ͨ������Na2O2����O2���������3���
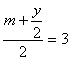 ��Ϊx��m ��������6-
��Ϊx��m ��������6-![]()
���۵���̬����x=3ʱ y��6��x=4ʱ y��4
���ݢ٢�������ͬʱ���㣬�Ӷ���C4H4����C3H6��
���Ը����Ŀ�����ΪC3H8��C4H6��
�ٸ���![]() ��3��
��3��
y=8ʱ��m=2 ![]() =
=![]()
y=6ʱ��m=3 ![]() =
=![]()



| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
��8�֣�(1998���Ϻ�)�ȼ��ⱥ��ʳ��ˮ��ȡNaOH�Ĺ�������ʾ��ͼ����
������ͼ�����������գ�
(1)�ڵ������У����Դ���������ĵ缫����������Ӧ�Ļ�ѧ����ʽΪ �����Դ���������ĵ缫��������ҺpH (ѡ����䡱�����ߡ����½���)��
(2)��ҵʳ�κ�Ca2+��Mg2+�����ʡ����ƹ��̷�����Ӧ�����ӷ���ʽΪ
�� ��
(3)���������SO42-�����ϸߣ��������ӱ��Լ���ȥSO42-���ñ��Լ�����
�� (ѡ��a��b��c��ѡ�۷�)��
a��Ba(OH)2 b��Ba(NO3)2 c��BaCl2
(4)Ϊ��Ч��ȥCa2+��Mg2+��SO42���������Լ��ĺ���˳��Ϊ (ѡ��a��b��c��ѡ�۷�)
a���ȼ�NaOH,���Na2CO3���ټӱ��Լ�
b���ȼ�NaOH����ӱ��Լ����ټ�Na2CO3
c���ȼӱ��Լ������NaOH���ټ�Na2CO3
(5)���ι���������NaOH��NaCl���ܽ���ϵIJ��죬ͨ�� ����ȴ�� (��д��������)��ȥNaCl
(6)�ڸ�Ĥ�����ʳ��ˮʱ�����۷ָ�Ϊ������������������ֹCl2��NaOH��Ӧ��������Ĥ������ʳ��ˮʱ��Cl2��NaOH��ֽӴ����������NaClO��H2����Ӧ�Ļ�ѧ����ʽΪ ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ʡ2011�������ѧһ�ֹ��ز��ԣ�7�� ���ͣ������
��8�֣�(1998���Ϻ�)�ȼ��ⱥ��ʳ��ˮ��ȡNaOH�Ĺ�������ʾ��ͼ����
������ͼ�����������գ�
(1)�ڵ������У����Դ���������ĵ缫����������Ӧ�Ļ�ѧ����ʽΪ �����Դ���������ĵ缫��������ҺpH (ѡ����䡱�����ߡ����½���)��
(2)��ҵʳ�κ�Ca2+��Mg2+�����ʡ����ƹ��̷�����Ӧ�����ӷ���ʽΪ
�� ��
(3)���������SO42-�����ϸߣ��������ӱ��Լ���ȥSO42-���ñ��Լ�����
�� (ѡ��a��b��c��ѡ�۷�)��
a��Ba(OH)2 b��Ba(NO3)2 c��BaCl2
(4)Ϊ��Ч��ȥCa2+��Mg2+��SO42���������Լ��ĺ���˳��Ϊ (ѡ��a��b��c��ѡ�۷�)
a���ȼ�NaOH,���Na2CO3���ټӱ��Լ�
b���ȼ�NaOH����ӱ��Լ����ټ�Na2CO3
c���ȼӱ��Լ������NaOH���ټ�Na2CO3
(5)���ι���������NaOH��NaCl���ܽ���ϵIJ��죬ͨ�� ����ȴ�� (��д��������)��ȥNaCl
(6)�ڸ�Ĥ�����ʳ��ˮʱ�����۷ָ�Ϊ������������������ֹCl2��NaOH��Ӧ��������Ĥ������ʳ��ˮʱ��Cl2��NaOH��ֽӴ����������NaClO��H2����Ӧ�Ļ�ѧ����ʽΪ ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ʡ2010-2011ѧ�������ѧһ�ֹ��ز��ԣ�7�� ���ͣ������
��8�֣�(1998���Ϻ�)�ȼ��ⱥ��ʳ��ˮ��ȡNaOH�Ĺ�������ʾ��ͼ����

������ͼ�����������գ�
(1)�ڵ������У����Դ���������ĵ缫����������Ӧ�Ļ�ѧ����ʽΪ �����Դ���������ĵ缫��������ҺpH (ѡ����䡱�����ߡ����½���)��
(2)��ҵʳ�κ�Ca2+��Mg2+�����ʡ����ƹ��̷�����Ӧ�����ӷ���ʽΪ
�� ��
(3)���������SO42-�����ϸߣ��������ӱ��Լ���ȥSO42-���ñ��Լ�����
�� (ѡ��a��b��c��ѡ�۷�)��
a��Ba(OH)2 b��Ba(NO3)2 c��BaCl2
(4)Ϊ��Ч��ȥCa2+��Mg2+��SO42���������Լ��ĺ���˳��Ϊ (ѡ��a��b��c��ѡ�۷�)
a���ȼ�NaOH,���Na2CO3���ټӱ��Լ�
b���ȼ�NaOH����ӱ��Լ����ټ�Na2CO3
c���ȼӱ��Լ������NaOH���ټ�Na2CO3
(5)���ι���������NaOH��NaCl���ܽ���ϵIJ��죬ͨ�� ����ȴ�� (��д��������)��ȥNaCl
(6)�ڸ�Ĥ�����ʳ��ˮʱ�����۷ָ�Ϊ������������������ֹCl2��NaOH��Ӧ��������Ĥ������ʳ��ˮʱ��Cl2��NaOH��ֽӴ����������NaClO��H2����Ӧ�Ļ�ѧ����ʽΪ ��
�鿴�𰸺ͽ���>>
����ѧУ��ѡ - ��ϰ���б� - �����б�
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com